Online Database of Chemicals from Around the World
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hong Xin Rui Yu Finechemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hbhxryfc.com | |||
 | +86 13886177946 | |||
 | +86 (27) 6552-6669 | |||
 | 2545103399@qq.com | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Wuhan Yuancheng Technology Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.yuanchengtechnology.com | |||
 | +86 17282536078 | |||
 | +86 17282536078 | |||
 | 3042184429@qq.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: 17282536078 | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Shaanxi Xi Yue Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xiyuezy.com | |||
 | +86 (029) 8823-0456 | |||
 | hcyhsbb@163.com | |||
| Chemical manufacturer since 1998 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Acros Organics | Belgium | |||
|---|---|---|---|---|
 | www.acros.com | |||
 | +86 (21) 5258-1100 | |||
 | +86 (21) 5258-0119 | |||
 | info@acros.com | |||
| Chemical manufacturer | ||||
| AK Scientific, Inc | USA | |||
|---|---|---|---|---|
 | www.aksci.com | |||
 | +1 (510) 429-8835 | |||
 | +1 (510) 429-8836 | |||
 | sales@aksci.com | |||
| Chemical manufacturer | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| Hubei Teyer Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hubeiteyer.com | |||
 | +86 (728) 533-2622 | |||
 | info@hubeiteyer.com | |||
| Chemical manufacturer since 2002 | ||||
| Jurong Renxin Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jrxchem.com | |||
 | +86 (519) 8222-0712 +86 13901490712 | |||
 | +86 (519) 8821-8921 | |||
 | sales@jrxchem.com | |||
| Chemical manufacturer | ||||
| Acanthus Research | USA | |||
|---|---|---|---|---|
 | www.acanthusresearch.com | |||
 | +1 (416) 978-4344 | |||
 | +1 (905) 566-9541 | |||
 | inquiries@acanthusresearch.com | |||
| Chemical manufacturer | ||||
| Natland International Corporation | USA | |||
|---|---|---|---|---|
 | www.natland.com | |||
 | +1 (919) 380-9775 | |||
 | +1 (919) 380-9886 | |||
 | info@natland.com | |||
| Chemical manufacturer | ||||
| Siegfried (USA), Inc. | USA | |||
|---|---|---|---|---|
 | www.siegfried.ch | |||
 | +1 (877) 763-8630 | |||
 | +1 (856) 678-8201 | |||
 | info@siegfried-usa.com | |||
| Chemical manufacturer | ||||
| Aronis | Russia | |||
|---|---|---|---|---|
 | www.aronis.ru | |||
 | +7 (926) 578-0336 | |||
 | +7 (495) 426-5336 | |||
 | rakishev@aronis.ru | |||
| Chemical manufacturer | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Classification | API >> Circulatory system medication >> Prevention and treatment of angina pectoris |
|---|---|
| Name | Nifedipine |
| Synonyms | Dimethyl 1,4-dihydro-2,6-dimethyl-4-(2'-nitrophenyl)-3,5-pyridinedicarboxylate; 1,4-Dihydro-2,6-dimethyl-4-(2-nitrophenyl)-3,5-pyridinedicarboxylic acid dimethyl ester |
| Molecular Structure | 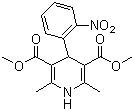 |
| Molecular Formula | C17H18N2O6 |
| Molecular Weight | 346.34 |
| CAS Registry Number | 21829-25-4 |
| EC Number | 244-598-3 |
| SMILES | CC1=C(C(C(=C(N1)C)C(=O)OC)C2=CC=CC=C2[N+](=O)[O-])C(=O)OC |
| Solubility | 69 mg/mL (DNSO), <1mg/mL (water) (Expl.) |
|---|---|
| Density | 1.3±0.1 g/cm3, Calc.* |
| Melting point | 174 °C (Expl.) |
| Index of Refraction | 1.559, Calc.* |
| Boiling Point | 475.3±45.0 °C (760 mmHg), Calc.* |
| Flash Point | 241.2±28.7 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS08 Warning Details GHS07;GHS08 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H361 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P264-P270-P280-P301+P317-P318-P330-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Nifedipine is a dihydropyridine calcium channel blocker that was first synthesized in the late 1960s as part of research into cardiovascular drugs. It was developed for its ability to inhibit calcium ion influx into vascular smooth muscle and myocardial cells, leading to vasodilation and reduced blood pressure. Nifedipine was introduced into clinical use in the 1970s and has since become an important medication in the treatment of hypertension and angina pectoris. As a calcium channel blocker, nifedipine selectively targets L-type calcium channels, preventing calcium-dependent contraction of vascular smooth muscle. This action results in arterial vasodilation, reducing peripheral resistance and lowering blood pressure. Additionally, nifedipine's ability to relax coronary arteries improves oxygen supply to the heart, making it effective in the management of angina. The drug is classified into immediate-release and extended-release formulations, with the latter designed to maintain stable plasma concentrations and minimize fluctuations in blood pressure. Nifedipine is widely prescribed for the treatment of hypertension, particularly in patients who require long-term blood pressure management. It is also used for the treatment and prevention of chronic stable angina and variant angina, conditions characterized by reduced coronary blood flow. In some cases, nifedipine has been employed in obstetrics to manage preterm labor due to its smooth muscle relaxant properties. However, its use in this context is subject to specific clinical guidelines. The pharmacokinetics of nifedipine include rapid absorption following oral administration, with metabolism occurring primarily in the liver through the cytochrome P450 system. The drug's elimination is predominantly renal, with inactive metabolites excreted in the urine. Due to its pharmacological profile, interactions with other medications that affect liver enzymes, such as certain antifungals and anticonvulsants, can influence its efficacy and safety. Nifedipine remains a key medication in cardiovascular therapy, with continued research focusing on its long-term effects and potential new applications. Advances in formulation technology have contributed to improved drug delivery, ensuring sustained efficacy with reduced side effects. References 2025. [Elevated blood pressure and hypertension : Focus of the 2024 ESC guidelines on risk reduction]. Herz, 50(1). DOI: 10.1007/s00059-024-05285-8 2025. Live-Cell Calcium Imaging in 3D Intestinal Organoids. Methods in molecular biology (Clifton, N.J.), 2792. DOI: 10.1007/978-1-0716-4164-4_16 2024. Severe influenza A viral pneumonia in a hemodialysis patient: successful treatment with steroid pulse therapy. CEN Case Reports, 13(6). DOI: 10.1007/s13730-024-00951-6 |
| Market Analysis Reports |