Online Database of Chemicals from Around the World
| Biosynth AG. | Switzerland | |||
|---|---|---|---|---|
 | www.biosynth.com | |||
 | +41 (71) 858-2020 | |||
 | +41 (71) 858-2030 | |||
 | welcome@biosynth.ch | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Zhejiang Arts & Crafts Imp & Exp.co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zjartspharm.com | |||
 | +86 571 87238903 | |||
 | jocelynpan@zjarts.com | |||
| Chemical distributor since 1992 | ||||
| chemBlink Standard supplier since 2024 | ||||
| IOLITEC Ionic Liquids Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.iolitec-usa.com | |||
 | +1 (205) 348-2831 | |||
 | +1 (205) 348-3510 | |||
 | info@iolitec-usa.com | |||
| Chemical manufacturer since 2002 | ||||
| Classification | Chemical reagent >> Organic reagent >> Sulfonate / sulfinate |
|---|---|
| Name | 1-Butyl-1-methylpyrrolidinium bistriflimide |
| Synonyms | Butylmethylpyrrolidinium bis(trifluoromethanesulfonyl)imide; Butylmethylpyrrolidinium bis(trifluoromethylsulfonyl)imide; N,N-Butylmethylpyrrolidinium bis(trifluoromethanesulfonyl)amide; N,N-Butylmethylpyrrolidinium bis(trifluoromethanesulfonyl)imide |
| Molecular Structure | 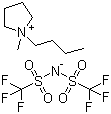 |
| Molecular Formula | C9H20N.C2F6NO4S2 |
| Molecular Weight | 422.41 |
| CAS Registry Number | 223437-11-4 |
| EC Number | 606-989-0 |
| SMILES | CCCC[N+]1(CCCC1)C.C(F)(F)(F)S(=O)(=O)[N-]S(=O)(=O)C(F)(F)F |
| Melting point | -18 °C |
|---|---|
| Refractive index | 1.4216 |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
1-Butyl-1-methylpyrrolidine bistrifluorocarbonimide (BMpyrrolidinium NTf₂) is a special class of ionic liquids known for its unique properties and diverse applications. This compound, with the chemical formula C₁₂H₁₈N₂O₄S₂F₆, represents a subclass of ionic liquids that have attracted attention for their unique combination of stability, conductivity, and versatility. The development of BMpyrrolidinium NTf₂ is part of a broader research program to explore ionic liquids with tailored properties. These studies began in the late 20th and early 21st century, when scientists were searching for solvents with low volatility, high thermal stability, and excellent ionic conductivity. The synthesis of BMpyrrolidinium NTf₂ was done to investigate new combinations of cations and anions, taking advantage of the favorable properties of the bistrifluorocarbonimide (NTf₂) anion. The synthesis generally involves the reaction of 1-butyl-1-methylpyrrolidine chloride with lithium bistrifluorocarbonimide. 1-Butyl-1-methylpyrrolidine chloride is first synthesized from pyrrolidine, butyl halide, and a methylating agent, and 1-Butyl-1-methylpyrrolidine chloride reacts with lithium bistrifluorocarbonimide to produce BMpyrrolidinium NTf₂ and lithium chloride. BMpyrrolidinium NTf₂ is a highly stable and effective ionic liquid suitable for a variety of applications. BMpyrrolidinium NTf₂ is used in electrochemical devices due to its high ionic conductivity and stability. It can be used as an electrolyte in batteries, supercapacitors, and fuel cells. Its low viscosity and wide electrochemical window enhance the performance of these devices, helping to improve energy storage and conversion efficiency. As an ionic liquid, BMpyrrolidinium NTf₂ offers an environmentally friendly alternative to traditional solvents. It is non-volatile, reducing the risk of harmful emissions, making it suitable for sustainable chemical processes. BMpyrrolidinium NTf₂ is used in organic synthesis and extraction to improve reaction selectivity and minimize environmental impact. BMpyrrolidinium NTf₂ is effective as a solvent and co-catalyst in a variety of catalytic reactions. Its stability and solvation properties make it a valuable medium in homogeneous and heterogeneous catalysis. Ionic liquids can increase reaction rates and yields and provide a robust environment for catalytic processes. In separation processes, BMpyrrolidinium NTf₂ is used in liquid-liquid extraction and ion exchange. Its ability to selectively dissolve and separate specific compounds makes it useful for chemical purification and recovery of valuable materials. This application is particularly valuable in industries that require precise separation and purification techniques. BMpyrrolidinium NTf₂ is used in materials science to synthesize and modify advanced materials. It is used to prepare nanoparticles and polymers, where it acts as a solvent or stabilizer. The properties of ionic liquids affect the size, shape, and stability of nanomaterials, helping to develop new materials with tailored properties. References 2024. Molecular Mobility Study of 1-Butyl-1-Methylpyrrolidinium Bis(trifluoromethylsulfonyl)imide Ionic Liquid by NMR Diffusometry. *Applied Magnetic Resonance*, 45(7). DOI: 10.1007/s00723-024-01678-4 2024. Temperature and Alkyl Chain Dependence of Physicochemical Properties of Pyrrolidinium- and Imidazolium-Based Ionic Liquids. *Journal of Solution Chemistry*, 53(2). DOI: 10.1007/s10953-023-01358-0 2009. Sol-gel synthesis of alumina, titania and mixed alumina/titania in the ionic liquid 1-butyl-1-methylpyrrolidinium bis(trifluoromethylsulphonyl) amide. *Journal of Materials Science*, 44(1). DOI: 10.1007/s10853-008-3107-y |
| Market Analysis Reports |