Online Database of Chemicals from Around the World
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Papchem Lifesciences | India | |||
|---|---|---|---|---|
 | papchemlifesciences.com | |||
 | 9061328401 | |||
 | bd@papchemlifesciences.com | |||
| Chemical manufacturer since 2021 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Apollo Scientific Ltd. | UK | |||
|---|---|---|---|---|
 | www.apolloscientific.co.uk | |||
 | +44 (161) 406-0505 | |||
 | +44 (161) 406-0506 | |||
 | sales@apolloscientific.co.uk | |||
| Chemical manufacturer | ||||
| Anisyn, Inc. | USA | |||
|---|---|---|---|---|
 | www.anisyn.com | |||
 | +1 (269) 372-8736 | |||
 | +1 (269) 372-3397 | |||
 | sales@anisyn.com | |||
| Chemical manufacturer | ||||
| Chemos GmbH & Co. KG | Germany | |||
|---|---|---|---|---|
 | www.chemos.de | |||
 | +49 871-966346-0 | |||
 | +49 871-966346-13 | |||
 | chemos@chemos.de | |||
| Chemical distributor | ||||
| Organo Fine Chemicals | India | |||
|---|---|---|---|---|
 | www.organo.in | |||
 | +91 (22) 2201-6968 2206-7521 | |||
 | +91 (22) 2205-9299 / 2208-3184 | |||
 | info@organo.in | |||
| Chemical manufacturer | ||||
| Real-Times (Beijing) Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.real-times.com.cn | |||
 | +86 (10) 8259-8075 +86 13426307855 | |||
 | +86 (10) 8259-7807 | |||
 | real-times@163.com | |||
| Chemical manufacturer since 2006 | ||||
| Wako Pure Chemical Industries, Ltd. | Japan | |||
|---|---|---|---|---|
 | www.wako-chem.co.jp | |||
 | +81 (6) 6203-3741 | |||
 | +81 (6) 6201-5964 | |||
 | wkhk.info@fujifilm.com | |||
| Chemical manufacturer since 1922 | ||||
| Classification | Biochemical >> Carbohydrate >> Double sugar |
|---|---|
| Name | Alloxan monohydrate |
| Synonyms | 1,3-Diazinane-2,4,5,6-tetrone hydrate; Pyrimidine-2,4,5,6(1H,3H)-tetrone hydrate; Mesoxalylurea monohydrate |
| Molecular Structure | 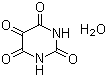 |
| Molecular Formula | C4H2N2O4.H2O |
| Molecular Weight | 160.08 |
| CAS Registry Number | 2244-11-3 |
| EC Number | 607-078-0 |
| SMILES | C1(=O)C(=O)NC(=O)NC1=O.O |
| Melting point | 240 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H332 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P270-P271-P280-P301+P317-P302+P352-P304+P340-P317-P321-P330-P362+P364-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Alloxan monohydrate, with the molecular formula C₄H₂O₄·H₂O, is recognized for its role in scientific research, particularly in studies related to diabetes and oxidative stress. Alloxan was first synthesized by German chemist Friedrich Wöhler in the 19th century while studying urea and related compounds. In 1839, Wöhler and his colleagues discovered that alloxan is a product of the oxidation of uric acid. Later, the monohydrate form, in which the compound is bound to one water molecule, was characterized and isolated, providing a better understanding of its chemical properties. Alloxan monohydrate is prepared by the oxidation of uric acid, typically by treating uric acid with potassium permanganate or other strong oxidizing agents to produce alloxan. The resulting alloxan is crystallized from an aqueous solution to form alloxan monohydrate. The synthesis produces a white crystalline solid that is soluble in water. Alloxan monohydrate is widely used in biomedical research to induce diabetes in experimental animals. It is selectively toxic to insulin-producing pancreatic beta cells, making it an effective tool for studying the pathogenesis of diabetes and testing potential treatments. Researchers use alloxan to create animal models of diabetes, which aids in understanding the disease and developing new treatments. The compound is also used to study oxidative stress and its effects on biological systems. Alloxan induces oxidative damage by producing reactive oxygen species (ROS), providing insights into how cells respond to oxidative stress. These studies have helped understand the mechanisms of various diseases, including neurodegenerative diseases and cancer. In organic synthesis, alloxan monohydrate serves as a reagent for a variety of chemical reactions. It is used to produce derivatives and complex molecules, aiding in the development of new compounds with potential applications in pharmaceuticals and materials science. Alloxan is used in analytical chemistry to study the reactivity and properties of different compounds. It is used as a standard in assays and as a reference material for various analytical techniques, aiding in the development of methods to detect and quantify chemicals. While alloxan is not widely used as a therapeutic agent, its role in research has led to studies of its potential as a drug. Studies have explored its effects on glucose metabolism and its interactions with cellular systems, aiding in a broader understanding of metabolic disorders and potential therapeutic interventions. References 2024. Integration of texture analysis based on DCE-MRI Ktrans map and metabolomics of early bone marrow microvascular changes in alloxan-induced diabetic rabbits. *BMC Medical Imaging*, 24(1). DOI: 10.1186/s12880-024-01416-z 2023. Antidiabetic activity of mango peel extract and mangiferin in alloxan-induced diabetic rats. *Future Journal of Pharmaceutical Sciences*, 9(1). DOI: 10.1186/s43094-023-00472-6 1966. Kidney lesions in rats with severe long-term alloxan diabetes: I. Influence of age, alloxan damage, and insulin administration. *Diabetologia*, 2(1). DOI: 10.1007/bf01257908 |
| Market Analysis Reports |