Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Shanghai ZaiQi Bio-Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemzq.com | |||
 | +86 (21) 5482-4098 | |||
 | +86 (21) 5482-4069 | |||
 | sales1@chemzq.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Nanjing Chemfly Medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemfly.com | |||
 | +86 18910986335 | |||
 | info@chemfly.com | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Intatrade Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.intatrade.de | |||
 | +49 (3493) 605-465 | |||
 | +49 (3493) 605-470 | |||
 | sales@intatrade.de | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Finetech Industry Limited | China | |||
|---|---|---|---|---|
 | www.finetechchem.com | |||
 | +86 (27) 8746-5837 +86 18971612321 | |||
 | +86 (27) 8777-2287 | |||
 | sales@finetechnology-ind.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Atlantic Research Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.atlantic-chemicals.com | |||
 | +44 (8707) 746-454 | |||
 | +44 (8707) 746-455 | |||
 | info@atlantic-chemicals.com | |||
| Chemical manufacturer | ||||
| Carbone Scientific Co., Ltd. | UK | |||
|---|---|---|---|---|
 | www.carbonesci.com | |||
 | +44 (870) 486-8629 | |||
 | +44 (870) 288-7399 | |||
 | sales@carbonesci.com | |||
| Chemical distributor | ||||
| Anisyn, Inc. | USA | |||
|---|---|---|---|---|
 | www.anisyn.com | |||
 | +1 (269) 372-8736 | |||
 | +1 (269) 372-3397 | |||
 | sales@anisyn.com | |||
| Chemical manufacturer | ||||
| Matrix Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.matrixscientific.com | |||
 | +1 (803) 788-9494 | |||
 | +1 (803) 788-9419 | |||
 | sales@matrixscientific.com | |||
| Chemical manufacturer | ||||
| Activate Scientific GmbH | Germany | |||
|---|---|---|---|---|
 | www.activate-scientific.com | |||
 | +49 (8051) 965-1660 | |||
 | +49 (8051) 965-1661 | |||
 | rschmitt@activate-scientific.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic aldehyde (containing acetal, hemiacetal) |
|---|---|
| Name | 4-Bromo-2-hydroxybenzaldehyde |
| Molecular Structure | 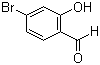 |
| Molecular Formula | C7H5BrO2 |
| Molecular Weight | 201.02 |
| CAS Registry Number | 22532-62-3 |
| EC Number | 636-586-5 |
| SMILES | C1=CC(=C(C=C1Br)O)C=O |
| Density | 1.7±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 50 - 54 °C (Expl.) |
| Boiling point | 256.2±25.0 °C 760 mmHg (Calc.)* |
| Flash point | 108.8±23.2 °C (Calc.)*, 110 °C (Expl.) |
| Index of refraction | 1.657 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS09 Warning Details GHS07;GHS09 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H319-H400 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P264-P264+P265-P270-P273-P280-P301+P317-P305+P351+P338-P330-P337+P317-P391-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
4‑Bromo‑2‑hydroxybenzaldehyde (also called 4-bromosalicylaldehyde) is an aromatic aldehyde with the molecular formula C7H5BrO2 and CAS number 22532‑62‑3. It is typically a pale‑yellow to off‑white crystalline solid, melting around 50–54 °C. It is used primarily as an intermediate in organic synthesis. The presence of both a bromine atom (at the 4‑position) and a hydroxyl group (at the 2‑position) makes it a versatile building block for further functionalization: the bromo substituent is suitable for cross‑coupling reactions (e.g., Suzuki, Heck) to introduce various aryl or alkenyl groups, while the aldehyde can participate in condensation, reduction, or further derivatization. The hydroxy group provides potential for intramolecular hydrogen bonding or for transformation (e.g., O-alkylation), which can influence reactivity and downstream synthetic pathways. In medicinal chemistry, derivatives of 4-bromo-2-hydroxybenzaldehyde are employed in the design of biologically active molecules, including enzyme inhibitors, fluorescent probes, and metal-chelating ligands. Its dual functionality allows chemists to access molecules that combine aromatic, donor, and reactive groups in a single scaffold. It has also been used in dye and material‑science chemistry, where the aromatic aldehyde can be converted into Schiff bases, polymerizable monomers, or chromophores. From a practical standpoint, the compound is stored under dry conditions and often protected from light to prevent decomposition. In lab reactions, it is usually dissolved in polar organic solvents like methanol or dichloromethane, and standard safety procedures must be followed given the potentially toxic nature of brominated aromatic aldehydes. As for synthesis, it can be prepared by bromination of salicylaldehyde or related phenolic precursors, followed by appropriate oxidation or formylation. The reactive aldehyde and phenolic groups must often be protected during bromination to ensure regioselectivity and yield. Because of its reactivity and functional group diversity, 4‑bromo‑2‑hydroxybenzaldehyde remains a valuable intermediate in research labs focused on synthetic organic chemistry, medicinal chemistry, and materials science. References Wang Y, Li H, Chen Q, et al. (2025) Detection of Latent Fingerprints with Simple AIE-Active p-Phenylenediamine Schiff Bases. Journal of Fluorescence 35 5 403–420 DOI: 10.1007/s10895-025-04339-2 Zhang X, Liu J, Wang P, et al. (2025) Synthesis, Crystal Structures and Catalytic Oxidation Property of Copper(II) Complexes Derived from 5-Bromo-2-((2-diethylaminoethylimino)methyl)Phenol. Journal of Structural Chemistry 66 5 723–734 DOI: 10.1134/s0022476625050178 Ahmadi R, Mohammadi A, et al. (2024) Design, synthesis, characterization, and docking studies of hitherto unknown chlorinated thiazolidine, thiophene, and 2-iminochromene derivatives as protein-like protease inhibitors. Journal of the Iranian Chemical Society 21 7 2155–2169 DOI: 10.1007/s13738-024-03056-0 |
| Market Analysis Reports |