Online Database of Chemicals from Around the World
| Ningbo Wingoo Semiconductor Core Material Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.wingooxc.com | |||
 | +86 18842662104 | |||
 | zoulin@wingooxc.com | |||
| Chemical manufacturer since 2025 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | Organic raw materials >> Aryl compounds >> Naphthalenes |
|---|---|
| Name | 4-(2-Phenyl-2-propanyl)phenyl 3-diazo-4-oxo-3,4-dihydro-1-naphthalenesulfonate |
| Synonyms | 2-diazonio-4-[4-(2-phenylpropan-2-yl)phenoxy]sulfonylnaphthalen-1-olate |
| Molecular Structure | 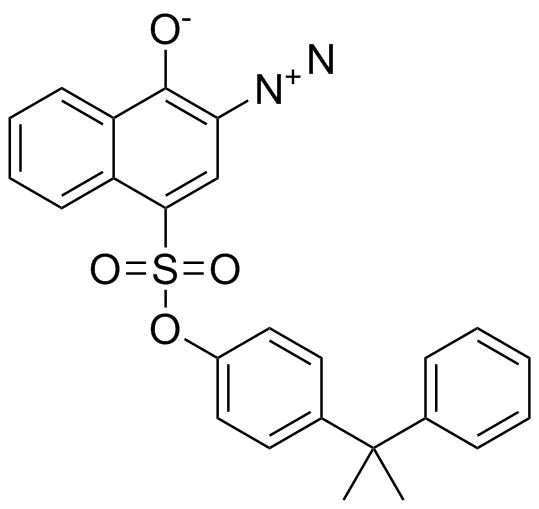 |
| Molecular Formula | C25H20N2O4S |
| Molecular Weight | 444.50 |
| CAS Registry Number | 23121-00-8 |
| EC Number | 694-898-7 |
| SMILES | CC(C)(C1=CC=CC=C1)C2=CC=C(C=C2)OS(=O)(=O)C3=CC(=C(C4=CC=CC=C43)[O-])[N+]#N |
| Hazard Classification | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||
|
4-(2-Phenyl-2-propanyl)phenyl 3-diazo-4-oxo-3,4-dihydro-1-naphthalenesulfonate belongs to a class of compounds derived from diazonaphthoquinone chemistry, which was developed and studied extensively during the growth of photoresist technology in the mid-20th century. The core structural motif of this compound is based on naphthoquinone derivatives bearing a diazo functional group, which was first investigated in the context of aromatic diazo chemistry in the 19th century following the discovery of diazonium salts and their reactivity with phenolic and aromatic systems. The combination of diazo chemistry with naphthoquinone scaffolds became particularly significant when researchers explored light-sensitive materials capable of undergoing controlled chemical transformations. The naphthalene-based quinone system in such compounds is known to exhibit strong absorption in the ultraviolet region, which enables photochemical activation of the diazo group. Early studies on naphthoquinone diazides showed that upon exposure to ultraviolet light, nitrogen gas is released from the diazo group, leading to the formation of highly reactive intermediates. This photochemical behavior was systematically studied in the context of developing positive photoresist materials for microfabrication. The sulfonate ester linkage in this compound further modifies its solubility characteristics and allows it to be incorporated into polymeric matrices used in coating formulations. The development of diazonaphthoquinone sulfonate esters is closely associated with the evolution of semiconductor manufacturing technology. As integrated circuits became more complex, there was a need for materials that could undergo precise spatial chemical changes under light exposure. Compounds such as 4-(2-phenyl-2-propanyl)phenyl 3-diazo-4-oxo-3,4-dihydro-1-naphthalenesulfonate were designed to meet these requirements by combining a light-sensitive diazo group with hydrophobic aromatic substituents that control dissolution behavior. The bulky 2-phenyl-2-propanyl substituent contributes to the physical properties of resist films, including film formation and resistance to unwanted dissolution before exposure. In practical application, diazonaphthoquinone-based sulfonate esters are widely used in positive photoresist systems. When exposed to ultraviolet radiation, the diazo group undergoes photolysis, releasing nitrogen gas and generating a ketene intermediate. This intermediate can react with water or other nucleophiles, resulting in a transformation that significantly increases the solubility of the exposed regions in alkaline developers. This selective solubility change allows for the transfer of fine patterns onto semiconductor substrates, which is a fundamental step in photolithographic fabrication processes. The use of these compounds has been central to the advancement of microelectronics. Their ability to provide high-resolution patterning has made them essential in the production of integrated circuits, printed circuit boards, and microelectromechanical systems. Over time, modifications to the substituent groups on the naphthalene and phenyl rings have been introduced to optimize sensitivity, resolution, and process stability. The incorporation of sulfonate ester functionality allows fine tuning of dissolution rates in developer solutions, which is critical for achieving precise control in lithographic processes. Beyond semiconductor applications, diazonaphthoquinone derivatives have also been applied in the field of microfabrication for producing microfluidic devices and patterned coatings. Their photochemical properties allow for the creation of complex structures without the need for mechanical processing. The development of such materials has contributed significantly to advancements in nanotechnology and precision engineering. The chemical study of these compounds continues to be relevant in materials science, particularly in the design of next-generation photoresists that require improved sensitivity and environmental stability. The fundamental chemistry of diazo group decomposition, naphthoquinone reactivity, and sulfonate ester modification remains an active area of research in understanding how molecular structure influences photochemical performance. Overall, 4-(2-phenyl-2-propanyl)phenyl 3-diazo-4-oxo-3,4-dihydro-1-naphthalenesulfonate represents an important example of a functional organic material developed from diazonaphthoquinone chemistry. Its discovery is closely tied to the evolution of photochemical research and its application is deeply embedded in modern microfabrication and semiconductor technology. References https://www.epa.gov/tsca-inventory |
| Market Analysis Reports |