Online Database of Chemicals from Around the World
| Discovery Fine Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.discofinechem.com | |||
 | +44 (1202) 874-517 | |||
 | +44 (845) 094-4385 | |||
 | pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Jurong Renxin Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jrxchem.com | |||
 | +86 (519) 8222-0712 +86 13901490712 | |||
 | +86 (519) 8821-8921 | |||
 | sales@jrxchem.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Yung Zip Chemical Co., Ltd. | Taiwan | |||
|---|---|---|---|---|
 | www.yungzip.com | |||
 | +886 (4) 2681-8780 2681-1674 | |||
 | +886 (4) 2681-2911 | |||
 | yspgyzc@yungshingroup.com | |||
| Chemical manufacturer since 1978 | ||||
| Classification | API >> Synthetic anti-infective drugs >> Antifungal drugs |
|---|---|
| Name | Econazole nitrate |
| Synonyms | 1-[2-[(4-Chlorophenyl)methoxy]-2-(2,4-dichlorophenyl)-ethyl]-1H-imidazole mononitrate; 1-(2,4-Dichloro-beta-((p-chlorobenzyl)oxy)phenethyl)imidazole nitrate |
| Molecular Structure | 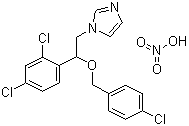 |
| Molecular Formula | C18H15Cl3N2O.HNO3 |
| Molecular Weight | 444.70 |
| CAS Registry Number | 24169-02-6 |
| EC Number | 246-053-5 |
| SMILES | C1=CC(=CC=C1COC(CN2C=CN=C2)C3=C(C=C(C=C3)Cl)Cl)Cl.[N+](=O)(O)[O-] |
| Melting point | 162 °C |
|---|---|
| Water solubility | <0.1 g/100 mL at 19 °C |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319 Details | ||||||||||||||||||||||||||||
| Safety Statements | P264-P264+P265-P270-P280-P301+P317-P302+P352-P305+P351+P338-P321-P330-P332+P317-P337+P317-P362+P364-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Econazole nitrate is a broad-spectrum antifungal agent that belongs to the imidazole class of synthetic antimycotic compounds. It is widely used in topical formulations for the treatment of fungal infections affecting the skin, nails, and mucous membranes. The compound consists of the active antifungal agent econazole, which is a substituted imidazole derivative, combined with nitric acid to form the nitrate salt, enhancing its solubility and stability for pharmaceutical use. The molecular structure of econazole includes a 1H-imidazole ring substituted at the 1-position with a 2,4-dichlorobenzyl group, and at the 2-position with an o-methoxyphenylethyl chain. These aromatic and alkyl substituents contribute to the compound’s lipophilicity, enabling it to penetrate fungal cell membranes effectively. The nitrate salt form, with the general formula C18H15Cl3N2O1·HNO3, is used in most medicinal preparations. Econazole nitrate was developed in the early 1970s and introduced into clinical use shortly thereafter. Its development was part of a larger effort to produce imidazole derivatives with improved antifungal properties and reduced toxicity compared to earlier antifungal agents. Since its introduction, econazole nitrate has become a widely used over-the-counter and prescription medication in dermatology and gynecology. The antifungal activity of econazole nitrate is primarily attributed to its inhibition of ergosterol biosynthesis, a key component of fungal cell membranes. Econazole binds to fungal cytochrome P450 enzymes, particularly lanosterol 14α-demethylase, preventing the conversion of lanosterol to ergosterol. This disruption results in increased membrane permeability, leakage of cellular contents, and ultimately cell death. Its selectivity for fungal cells over human cells arises from the fundamental differences between ergosterol and cholesterol biosynthesis. Econazole nitrate is effective against a variety of pathogenic fungi, including dermatophytes such as *Trichophyton*, *Epidermophyton*, and *Microsporum* species, as well as yeasts like *Candida albicans*. It also exhibits antibacterial activity against certain gram-positive bacteria, including *Staphylococcus aureus*, making it suitable for treating mixed infections. The compound is typically administered in topical formulations such as creams, powders, sprays, and vaginal suppositories. The usual concentration in commercial products is 1% econazole nitrate. Treatment regimens vary depending on the location and severity of the infection, but topical application is generally performed once or twice daily for one to four weeks. Adverse effects associated with econazole nitrate are usually mild and localized. They may include burning, itching, redness, or irritation at the site of application. Systemic absorption is minimal when used topically, reducing the risk of systemic side effects. However, caution is recommended in individuals with known hypersensitivity to imidazole compounds. Econazole nitrate is chemically stable under normal storage conditions and has a shelf life of several years in properly sealed containers. It is sensitive to strong oxidizers and should be stored in a cool, dry place, away from direct light and heat. In addition to its use in human medicine, econazole nitrate is occasionally used in veterinary formulations for treating fungal infections in animals. Its broad antifungal spectrum and established safety profile make it suitable for dermatological use across species. In conclusion, econazole nitrate is a well-established antifungal medication used primarily in topical treatments for superficial fungal infections. Its imidazole-based structure, combined with a nitrate counterion, provides potent antifungal activity through inhibition of ergosterol synthesis. Econazole nitrate remains an important therapeutic agent in both human and veterinary medicine due to its efficacy, safety, and versatility in formulation. References 2021. Structural mechanisms of TRPV6 inhibition by ruthenium red and econazole. Nature Communications, 12(1). DOI: 10.1038/s41467-021-26608-x 2021. Formulation Development, In Vitro and In Vivo Evaluation of Topical Hydrogel Formulation of Econazole Nitrate-Loaded β-Cyclodextrin Nanosponges. Journal of Pharmaceutical Sciences, 110(11). DOI: 10.1016/j.xphs.2021.07.008 2020. Development and evaluation of a film-forming system hybridized with econazole-loaded nanostructured lipid carriers for enhanced antifungal activity against dermatophytes. Acta Biomaterialia, 101. DOI: 10.1016/j.actbio.2019.10.024 |
| Market Analysis Reports |