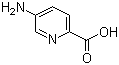
5-Amino-2-pyridinecarboxylic acid, also known as 5-amino-2-pyridinecarboxylate or 5-amino-2-carboxypyridine, is a chemical compound belonging to the class of pyridinecarboxylic acids. The compound has the molecular formula C6H6N2O2 and consists of a pyridine ring with both an amino group and a carboxyl group attached at the 5- and 2-positions, respectively.
The discovery and synthesis of 5-amino-2-pyridinecarboxylic acid can be traced to the development of pyridine-based intermediates and their increasing use in synthetic organic chemistry. Pyridine derivatives, including substituted pyridinecarboxylic acids, have long been of interest due to their wide range of applications in pharmaceuticals, agrochemicals, and materials science. The specific synthesis of 5-amino-2-pyridinecarboxylic acid typically involves standard methods of organic synthesis, including functionalization of the pyridine ring through nitration and subsequent reduction steps.
5-Amino-2-pyridinecarboxylic acid is primarily used as an intermediate in the synthesis of biologically active compounds. It serves as a valuable building block in the development of drugs and other bioactive molecules. The amino group in the compound allows for further functionalization, such as the formation of amides or the introduction of additional substituents on the pyridine ring. This versatility has led to its use in the synthesis of various pharmaceutical agents with therapeutic potential. Its ability to form stable coordination complexes with metal ions is also of particular interest in medicinal chemistry, where metal-containing drugs are often employed.
In addition to its role in drug development, 5-amino-2-pyridinecarboxylic acid has been investigated for use in the creation of chelating agents. The compound's structure allows it to coordinate with metal ions, making it suitable for the development of chelators, which are useful in various applications, including metal extraction, environmental remediation, and as contrast agents in imaging technologies.
In terms of industrial applications, 5-amino-2-pyridinecarboxylic acid can be used in the synthesis of agrochemicals. The compound can be further modified to produce herbicides or fungicides, which benefit from the pyridine backbone's biological activity. Additionally, 5-amino-2-pyridinecarboxylic acid and its derivatives have been studied in the context of corrosion inhibition, where they function as protective agents for metal surfaces, helping to prevent the degradation of materials in various industrial settings.
This compound has also shown potential in the field of analytical chemistry. Its ability to participate in complexation reactions with metal ions has been utilized in the design of analytical techniques, including spectrophotometric assays and chromatography. Through these methods, 5-amino-2-pyridinecarboxylic acid can be employed in the detection of metal ions or other chemical species in various sample matrices.
5-Amino-2-pyridinecarboxylic acid is generally available through commercial chemical suppliers, and it is often used by researchers in laboratories for the synthesis of other pyridine derivatives. Its known stability and reactivity make it a useful intermediate for a variety of chemical transformations. The compound's chemical properties, such as its solubility in water and polar solvents, as well as its acid-base behavior, are well-documented in the literature, facilitating its handling and use in laboratory and industrial applications.
The applications of 5-amino-2-pyridinecarboxylic acid reflect its significance in both research and industry. Its role as a precursor in the synthesis of more complex molecules, particularly in the pharmaceutical and chemical industries, underscores its importance as a versatile and valuable chemical building block. Through its ability to form stable complexes with metal ions and its utility in the synthesis of biologically active compounds, 5-amino-2-pyridinecarboxylic acid continues to be of interest across a variety of scientific and industrial fields.
References
2017. Covalent immobilization of dysprosium-based metal-organic chains on silicon-based polymer brush surfaces. New J. Chem., 41(15).
DOI: 10.1039/C7NJ00837E
2016. Novel anti-diabetic and luminescent coordination compounds based on vanadium. New J. Chem., 40(6).
DOI: 10.1039/C5NJ03537A
2010. Identification of a potent activator of Akt phosphorylation from a novel series of phenolic, picolinic, pyridino, and hydroxamic zinc(II) complexes. Journal of biological inorganic chemistry : JBIC : a publication of the Society of Biological Inorganic Chemistry, 15(8).
DOI: 10.1007/s00775-010-0716-0
|











































































 GHS07 Warning Details
GHS07 Warning Details