Online Database of Chemicals from Around the World
| Epochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.epochem.com | |||
 | +86 (21) 6760-1595 6760-1597 | |||
 | +86 (21) 6760-1605 | |||
 | seth_wang@epochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Jinan Welt Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jinandayuchem.com | |||
 | +86 (531) 8715-3728 8796-0445 8792-2518 8715-1180 | |||
 | +86 (531) 8792-2099 | |||
 | jndayu@163.com xufeng192@126.com feng192@jinandayuchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Wako Pure Chemical Industries, Ltd. | Japan | |||
|---|---|---|---|---|
 | www.wako-chem.co.jp | |||
 | +81 (6) 6203-3741 | |||
 | +81 (6) 6201-5964 | |||
 | wkhk.info@fujifilm.com | |||
| Chemical manufacturer since 1922 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridine compound >> Pyridine derivative |
|---|---|
| Name | 4-Pyrrolidinopyridine |
| Synonyms | 4-(Pyrrolidin-1-yl)pyridine |
| Molecular Structure | 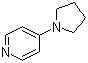 |
| Molecular Formula | C9H12N2 |
| Molecular Weight | 148.21 |
| CAS Registry Number | 2456-81-7 |
| EC Number | 219-534-2 |
| SMILES | C1CCN(C1)C2=CC=NC=C2 |
| Melting point | 54-58 °C |
|---|---|
| Boiling point | 171-173 °C (20.2527 mmHg) |
| Flash point | 142 °C |
| Water solubility | 3 g/L (21 °C) |
| Hazard Symbols |   GHS05;GHS06 Danger Details GHS05;GHS06 Danger Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H314 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P260-P264-P270-P280-P301+P316-P301+P330+P331-P302+P361+P354-P304+P340-P305+P354+P338-P316-P321-P330-P363-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| Transport Information | UN 2923 | ||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
4-Pyrrolidinopyridine, also known as 4-PPY, was discovered in the late 1960s during research into heterocyclic compounds. It was synthesized through the reaction between pyridine and pyrrolidine, resulting in the formation of a five-membered heterocycle containing both pyridine and pyrrolidine moieties. The discovery of 4-Pyrrolidinopyridine marked a significant advancement in organic chemistry, providing chemists with a versatile building block for the synthesis of various compounds. 4-Pyrrolidinopyridine is widely used as a catalyst in organic synthesis reactions. Its unique structure and catalytic properties make it suitable for a variety of transformations, including acylation, alkylation, and condensation reactions. As a base catalyst, 4-Pyrrolidinopyridine facilitates the formation of carbon-carbon and carbon-nitrogen bonds, enabling the synthesis of complex molecules in a more efficient and selective manner. In the pharmaceutical industry, 4-Pyrrolidinopyridine serves as a valuable intermediate for the synthesis of pharmaceutical compounds. Its heterocyclic structure and reactivity make it a versatile building block for the production of various drugs, including antivirals, antibiotics, and antihypertensives. Chemical modifications of the pyrrolidine and pyridine rings enable the creation of drug candidates with improved pharmacological properties, such as increased potency, selectivity, and metabolic stability. 4-Pyrrolidinopyridine is utilized as a catalyst in polymerization reactions, particularly in the synthesis of polyamides and polyesters. Its catalytic activity facilitates the polymerization of monomers, leading to the formation of high molecular weight polymers with desired properties such as thermal stability, mechanical strength, and biodegradability. 4-Pyrrolidinopyridine-based catalysts are employed in various polymerization processes, including ring-opening polymerization and condensation polymerization, for the production of polymers used in industries such as textiles, plastics, and biomedical materials. In organic synthesis and laboratory research, 4-Pyrrolidinopyridine is commonly used as a solvent and reagent. Its solubility properties and compatibility with a wide range of organic and inorganic compounds make it useful for dissolving or reacting with other substances to form desired products. 4-Pyrrolidinopyridine is employed in various synthetic transformations, such as acylation, alkylation, and cyclization reactions, where its role as a base or nucleophile facilitates the formation of new chemical bonds. In electrochemical applications, 4-Pyrrolidinopyridine is utilized as an electrolyte additive in batteries and capacitors. Its high solubility in common electrolyte solvents and ability to form stable complexes with metal ions contribute to improved electrolyte conductivity and electrochemical performance. 4-Pyrrolidinopyridine-based electrolytes are employed in lithium-ion batteries, supercapacitors, and other energy storage devices, enhancing their efficiency, cycle life, and safety. References 2016. Catalytic enantioselective acyl transfer: the case for 4-PPY with a C-3 carboxamide peptide auxiliary based on synthesis and modelling studies. *Organic & Biomolecular Chemistry*, 14(46). DOI: 10.1039/c6ob01991a 2016. DFT analysis of the nucleophilicity of substituted pyridines and prediction of new molecules having nucleophilic character stronger than 4-pyrrolidino pyridine. *Journal of Chemical Sciences*, 128(3). DOI: 10.1007/s12039-016-1057-5 1979. A convenient synthesis of phosphatidylcholines: acylation of sn-glycero-3-phosphocholine with fatty acid anhydride and 4-pyrrolidinopyridine. *Journal of Lipid Research*, 20(5). URL: https://pubmed.ncbi.nlm.nih.gov/490045 |
| Market Analysis Reports |