Online Database of Chemicals from Around the World
| Tianjin Pharmacn Medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharmacn.com | |||
 | +86 (22) 8372-6121 | |||
 | +86 (22) 2337-7103 | |||
 | marketing@pharmacn.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Biocompounds Pharmaceutical Inc. | China | |||
|---|---|---|---|---|
 | www.biocompounds.com | |||
 | +86 (21) 5768-7675 | |||
 | +86 (21) 5768-7169 | |||
 | sales@biocompounds.com info@biocompounds.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Manus Aktteva Biopharma Llp | China | |||
|---|---|---|---|---|
 | www.manusaktteva.com | |||
 | +91 ((79) 2646-3395 | |||
 | +91 ((79) 2646-3395 | |||
 | staff@manusakttevabiopharma.in | |||
| Chemical distributor since 2010 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Manus Aktteva | India | |||
|---|---|---|---|---|
 | www.manusaktteva.in | |||
 | +91 (79) 6512-3395 | |||
 | +91 (79) 2646-3395 | |||
 | products@manusakttevabiopharma.in | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2008 | ||||
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| KKyemistry (India) Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.kyemistryindia.com | |||
 | +91 (40) 2351-0950 | |||
 | +91 (40) 2351-0667 | |||
 | info@kyemistryindia.com | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shandong Boyuan Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.boyuanpharm.com | |||
 | +86 (531) 6995-4981 8896-3280 +86 15806417970 | |||
 | +86 (531) 8896-4879 | |||
 | Jeffrey.Liu@boyuanpharm.com boyuanchem@126.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Datang (Hangzhou) Pharmachem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.dtpharmachem.com | |||
 | +86 (571) 8731-1096 | |||
 | +86 (571) 8693-2703 | |||
 | 1609019596@qq.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Qichuang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qc-chemical.com | |||
 | +86 (571) 8893-5129 | |||
 | +86 (571) 8825-0182 | |||
 | david@qc-chemical.com sales@qc-chemical.com davidw0828@gmail.com | |||
 | QQ Chat | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Angene International Limited | Hong Kong | |||
|---|---|---|---|---|
 | www.angenechemical.com | |||
 | +852 8191-6999 | |||
 | +852 2723-7175 | |||
 | sales@angenechemical.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Jinan Aery Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.aerpharm.com | |||
 | +86 (531) 8126-3406 +86 15553134202 | |||
 | +86 (531) 5859-1634 | |||
 | sales@aerpharm.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Sanochem (Chengdu) Tech. Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sano-chem.com | |||
 | +86 15002851260 | |||
 | +86 (28) 8799-9436 | |||
 | sales@sano-chem.com | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Suli Pharmaceutical Technology Jiangyin Co., Ltd., | China | |||
|---|---|---|---|---|
 | www.sulipharma.com | |||
 | +86 (21) 5197-2823 ex 8033 | |||
 | qian.wang@suli.com | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Qufu Haida Tiancheng Biochemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hdtchg.com | |||
 | +86 (537) 4531868 +86 13345174153 | |||
 | qfhdtc@126.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Taizhou Creating Bio-pharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.creatingbiopharm.com | |||
 | +86 (0576) 8882-7176 | |||
 | +86 (0576) 8882-2496 | |||
 | post@creatingchemical.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: 18906581668 | |||
 | WhatsApp:+86 18906581668 | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2023 | ||||
| MolScanner | Singapore | |||
|---|---|---|---|---|
 | www.molscanner.com | |||
 | +86 18621675448 | |||
 | marketing@molscanner.com | |||
 | WhatsApp:9896 7603 | |||
| Chemical manufacturer since 2025 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Sinbond Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinbondpharm.com | |||
 | +86 (531) 8703-8285 +86 13583181986 +44 (208) 242-5518 | |||
 | +86 (531) 8703-8285 | |||
 | cici1124@gmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| Classification | Organic raw materials >> Amino compound >> Acyclic monoamines, polyamines and their derivatives and salts |
|---|---|
| Name | 3-Amino-2,6-piperidinedione hydrochloride |
| Synonyms | 2,6-Dioxopiperidine-3-ammonium chloride |
| Molecular Structure | 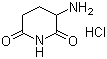 |
| Molecular Formula | C5H9ClN2O2 |
| Molecular Weight | 164.59 |
| CAS Registry Number | 24666-56-6 (2686-86-4) |
| EC Number | 807-866-6 |
| SMILES | C1CC(=O)NC(=O)C1N.Cl |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
3-Amino-2,6-piperidinedione hydrochloride is a chemical compound belonging to the class of piperidine derivatives, characterized by the presence of two carbonyl groups at the 2 and 6 positions on the piperidine ring, along with an amino group at the 3-position. The hydrochloride salt form of this compound makes it soluble in water, which is beneficial for various applications in research and pharmaceutical industries. The compound’s structure, combining a heterocyclic ring with amino and carbonyl functionalities, makes it a valuable intermediate for organic synthesis and medicinal chemistry. The synthesis of 3-amino-2,6-piperidinedione hydrochloride typically involves the functionalization of piperidine derivatives. The introduction of the amino group at the 3-position and the carbonyl groups at the 2 and 6 positions of the piperidine ring can be achieved through a variety of chemical methods, such as nucleophilic substitution or cyclization reactions. The hydrochloride salt form of the compound is generally prepared by reacting the free base with hydrochloric acid, which enhances the solubility and stability of the molecule in aqueous solutions. In terms of applications, 3-amino-2,6-piperidinedione hydrochloride has shown potential in several areas of medicinal chemistry due to its bioactive nature. The amino group on the molecule allows for further chemical modifications that can enhance its biological activity, making it a valuable lead structure for the development of pharmaceutical compounds. The presence of the two carbonyl groups in the piperidine ring also adds to the compound's reactivity, providing sites for further functionalization or interaction with biological targets, such as enzymes or receptors. One significant application of 3-amino-2,6-piperidinedione hydrochloride is in the development of central nervous system (CNS) drugs. Piperidine derivatives are commonly found in compounds that affect neurotransmitter systems, and this compound may have similar effects. The amino group can be involved in binding to neurotransmitter receptors or enzymes, possibly influencing the levels of important neurotransmitters like dopamine, serotonin, or norepinephrine. This makes 3-amino-2,6-piperidinedione hydrochloride a promising candidate for treating neurological disorders such as depression, anxiety, and neurodegenerative diseases like Alzheimer's or Parkinson's disease. Additionally, the compound’s structure allows it to participate in various chemical reactions that can lead to the creation of more complex molecules. Its ability to undergo cyclization and functional group transformations means that it can be used as an intermediate in the synthesis of other biologically active compounds. Researchers have explored the use of piperidinedione derivatives in drug discovery, particularly in the development of molecules that interact with GABA receptors, which play a key role in regulating nervous system activity. 3-amino-2,6-piperidinedione hydrochloride may also find applications in the synthesis of peptide-based drugs, where the piperidine ring can be incorporated into larger peptide chains to enhance stability, solubility, or bioavailability. The introduction of the amino group could help facilitate peptide bond formation, while the diketone functionality may serve as a reactive site for linking with other chemical entities. Beyond medicinal chemistry, 3-amino-2,6-piperidinedione hydrochloride may also have potential in the field of materials science. The ability of piperidine-based compounds to participate in various chemical transformations could make them useful in the synthesis of advanced organic materials, such as polymers or organic semiconductors. The compound’s reactivity and stability in aqueous environments can be leveraged in the development of functional materials with specific electronic or optical properties. In conclusion, 3-amino-2,6-piperidinedione hydrochloride is a versatile compound with applications in medicinal chemistry, particularly in the development of CNS-active drugs, and in the synthesis of more complex organic molecules. Its ability to undergo chemical transformations, coupled with its bioactive piperidine-based structure, makes it a valuable lead compound for drug discovery and materials science. The compound's potential in neuropharmacology and organic synthesis highlights its importance in various areas of chemical research and development. References 2023. Degradation of neurodegenerative disease-associated TDP-43 aggregates and oligomers via a proteolysis-targeting chimera. Journal of Biomedical Science, 30(1). DOI: 10.1186/s12929-023-00921-7 2022. Experimental design optimization for the synthesis of lenalidomide nitro precursor. Research on Chemical Intermediates, 48(11). DOI: 10.1007/s11164-022-04869-5 2018. Design and synthesis of novel bispecific molecules for inducing BRD4 protein degradation. Chemical Research in Chinese Universities, 34(1). DOI: 10.1007/s40242-018-7272-5 1998. Synthesis and antiaggregative activity of derivatives of 3-aminopiperidine-2,6-diones and 3-aminopyrrolidine-2,5-diones. Pharmaceutical Chemistry Journal, 32(6). DOI: 10.1007/bf02580514 |
| Market Analysis Reports |