Online Database of Chemicals from Around the World
| Xi'an Tuochao Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xatuochao.cn | |||
 | +86 18092957403 | |||
 | chen.zeshan@xatuochao.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: +8618092957403 | |||
 | WhatsApp:+8618092957403 | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Classification | Organic raw materials >> Aryl compounds |
|---|---|
| Name | Diphenyl(vinyl)sulfonium trifluoromethanesulfonate |
| Synonyms | ethenyl(diphenyl)sulfanium trifluoromethanesulfonate |
| Molecular Structure | 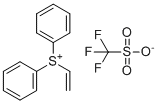 |
| Molecular Formula | C15H13F3O3S2 |
| Molecular Weight | 362.39 |
| CAS Registry Number | 247129-88-0 |
| SMILES | C=C[S+](C1=CC=CC=C1)C2=CC=CC=C2.C(F)(F)(F)S(=O)(=O)[O-] |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
Diphenyl(vinyl)sulfonium trifluoromethanesulfonate is an organosulfur compound that plays a significant role in modern synthetic chemistry. With the molecular formula C16H15F3O3S, it features a sulfonium ion derived from diphenylvinylsulfide, paired with a trifluoromethanesulfonate (or triflate) anion. This compound is particularly valued for its utility as a powerful electrophile in various organic transformations, including cross-coupling reactions and as a source of reactive intermediates. The discovery of diphenyl(vinyl)sulfonium trifluoromethanesulfonate can be traced back to the exploration of sulfonium salts in the mid-20th century. Researchers sought to develop reagents that could facilitate nucleophilic substitution reactions. The introduction of triflate as a leaving group significantly increased the reactivity of sulfonium compounds, allowing for a broader range of applications in synthetic organic chemistry. One of the most notable applications of diphenyl(vinyl)sulfonium trifluoromethanesulfonate is in the field of photochemistry. The compound can undergo photoinduced reactions, generating highly reactive carbocations that serve as intermediates in various photochemical processes. These intermediates can be utilized in the synthesis of complex organic molecules, making diphenyl(vinyl)sulfonium trifluoromethanesulfonate a valuable tool for chemists working in organic synthesis. In addition to its role in photochemical reactions, this compound has been employed as a versatile electrophile in the synthesis of vinyl compounds. The vinyl group in diphenyl(vinyl)sulfonium trifluoromethanesulfonate enables it to participate in nucleophilic substitutions, providing access to various functionalized products. Researchers have leveraged this reactivity to develop new methodologies for constructing complex molecular architectures. Moreover, diphenyl(vinyl)sulfonium trifluoromethanesulfonate has gained attention in the context of polymer chemistry. It can be utilized as a photoinitiator in the production of polymers through radical polymerization processes. The ability to generate radicals upon exposure to light makes this compound particularly useful in the development of advanced materials, including coatings, adhesives, and composites. This application aligns with the growing interest in photopolymerization techniques that enable the rapid and efficient production of polymeric materials. The environmental and safety aspects of using diphenyl(vinyl)sulfonium trifluoromethanesulfonate are important considerations in its application. As with many sulfonium compounds, proper handling and disposal methods are crucial to mitigate any potential hazards associated with its use. Researchers are continuously investigating safer alternatives and optimizing reaction conditions to enhance the sustainability of processes involving this compound. In summary, diphenyl(vinyl)sulfonium trifluoromethanesulfonate is a significant reagent in synthetic chemistry, particularly valued for its electrophilic properties and utility in photochemical processes. Its applications span organic synthesis, polymer chemistry, and the development of advanced materials. Ongoing research continues to explore its potential, ensuring its relevance in both academic and industrial settings. References 2013. New Entry to Polycyclic Fused Indoles via Gold(I)-catalyzed Cascade Reaction. Chemistry - An Asian Journal, 8(3). DOI: 10.1002/asia.201201249 2010. Remote Chiral Induction in Vinyl Sulfonium Salt-Mediated Ring Expansion of Hemiaminals into Epoxide-Fused Azepines. Chemistry - An Asian Journal, 6(1). DOI: 10.1002/asia.201000817 |
| Market Analysis Reports |