Online Database of Chemicals from Around the World
| BrightGene Bio-medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bright-gene.com | |||
 | +86 (512) 6255-1801 6255-1767 +86 13812696362 | |||
 | +86 (512) 6255-1799 | |||
 | kevinwan@bright-gene.com kevinwan0203@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Cherry Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cherrypharmatech.com | |||
 | +86 (571) 8163-6070 +86 18042403330 | |||
 | +86 (571) 8163-6070 | |||
 | info@cherrypharmatech.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Cangzhou Enke Pharma-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.enkepharma.com | |||
 | +86 (317) 510-5699 510-6597 +86 15533709196 | |||
 | +86 (317) 510-6596 | |||
 | sale@enkepharma.com enkepharma@126.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: ymzhao | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Changzhou Extraordinary Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.feifanchem.com | |||
 | +86 (519) 8211-1935 +86 15195055733 | |||
 | +86 (519) 8261-1250 | |||
 | info@feifanchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Cn Chemunion Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cnchemunion.com | |||
 | +86 17366116869 | |||
 | david@cnchemunion.com | |||
 | QQ Chat | |||
 | WeChat: +86 15250069576 | |||
 | WhatsApp:+86 17366116869 | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Sinova Corporation | USA | |||
|---|---|---|---|---|
 | www.sinovainc.com | |||
 | +1 (301) 961-1525 | |||
 | +1 (240) 235-4288 | |||
 | sales@sinovainc.com | |||
| Chemical manufacturer | ||||
| Classification | API >> Antineoplastic agents |
|---|---|
| Name | Eribulin |
| Synonyms | B 1939; E 7389; ER 086526 |
| Molecular Structure | 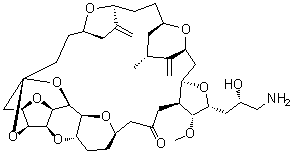 |
| Molecular Formula | C40H59NO11 |
| Molecular Weight | 729.90 |
| CAS Registry Number | 253128-41-5 |
| EC Number | 803-583-7 |
| SMILES | C[C@@H]1C[C@@H]2CC[C@H]3C(=C)C[C@@H](O3)CC[C@]45C[C@@H]6[C@H](O4)[C@H]7[C@@H](O6)[C@@H](O5)[C@@H]8[C@@H](O7)CC[C@@H](O8)CC(=O)C[C@H]9[C@H](C[C@H](C1=C)O2)O[C@@H]([C@@H]9OC)C[C@@H](CN)O |
| Solubility | Practically insoluble (0.015 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.29±0.1 g/cm3 (20 °C 760 Torr), Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2013 ACD/Labs) |
| Hazard Symbols |   GHS06;GHS08 Danger Details GHS06;GHS08 Danger Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301+H311+H331-H301-H311-H331-H341-H351-H361 Details | ||||||||||||||||||||||||||||
| Safety Statements | P203-P261-P262-P264-P270-P271-P280-P301+P316-P302+P352-P304+P340-P316-P318-P321-P330-P361+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Eribulin is a potent antineoplastic agent used primarily in the treatment of cancer. It is a synthetic analogue of halichondrin B, a natural product originally isolated from the marine sponge Halichondria okadai. Eribulin, with the chemical formula C17H23Br3O5, is notable for its unique mechanism of action and its role in treating various types of cancer, including metastatic breast cancer and liposarcoma. The discovery of eribulin began with research into natural marine products with potential therapeutic properties. Halichondrin B, a complex macrocyclic compound, was identified for its potent anticancer activity. However, the complex structure and challenging synthesis of halichondrin B prompted the development of synthetic analogues. Eribulin, synthesized by the chemical modification of halichondrin B, was designed to retain the key pharmacophoric elements responsible for its biological activity while simplifying the structure for more practical synthesis and application. Eribulin's primary application is in oncology. It is utilized as a treatment for patients with metastatic breast cancer who have previously received at least two other chemotherapeutic regimens for advanced disease. It is also used for the treatment of liposarcoma in patients who have already undergone prior treatments. The drug functions by inhibiting microtubule dynamics, a mechanism that disrupts cell division and effectively induces apoptosis in cancer cells. This action is achieved by binding to the tubulin protein, which stabilizes microtubules and prevents their disassembly, ultimately leading to mitotic arrest and cell death. The clinical efficacy of eribulin has been demonstrated in multiple studies. For metastatic breast cancer, it has shown improved overall survival and progression-free survival compared to other treatments. In liposarcoma, eribulin has provided significant benefits in terms of tumor control and patient outcomes. The drug's efficacy is often measured in terms of its ability to improve survival rates and delay disease progression in patients with advanced cancer stages. Beyond its primary uses, eribulin is also under investigation for potential applications in other cancer types and in combination therapies. Research is ongoing to explore its effectiveness in treating cancers beyond breast cancer and liposarcoma, as well as its potential synergistic effects when used in conjunction with other chemotherapeutic agents. References 2024. Immunomodulatory Effects of Halichondrin Isolated from Marine Sponges and Its Synthetic Analogs in Oncological Applications. Marine Drugs, 22(9). DOI: 10.3390/md22090426 2010. Phase II Study of the Halichondrin B Analog Eribulin Mesylate in Patients With Locally Advanced or Metastatic Breast Cancer Previously Treated With an Anthracycline, a Taxane, and Capecitabine. Journal of Clinical Oncology, 28(25). DOI: 10.1200/jco.2009.25.8467 2005. The primary antimitotic mechanism of action of the synthetic halichondrin E7389 is suppression of microtubule growth. Molecular Cancer Therapeutics, 4(7). DOI: 10.1158/1535-7163.mct-04-0345 |
| Market Analysis Reports |