Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Coresyn Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.coresyn.com | |||
 | +86 (571) 8662-6709 | |||
 | sales@coresyn.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Edu Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemido.com | |||
 | +86 19301251526 | |||
 | sales1@chemido.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Ryan Scientific, Inc. | USA | |||
|---|---|---|---|---|
 | www.ryansci.com | |||
 | +1 (843)-884-4911 | |||
 | +1 (843) 884-5568 | |||
 | sales@ryansci.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Triazines |
|---|---|
| Name | 1,3,5-Tribenzyl-1,3,5-triazinane |
| Synonyms | 1,3,5-Tribenzylhexahydro-1,3,5-triazine |
| Molecular Structure | 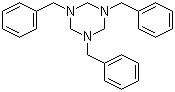 |
| Molecular Formula | C24H27N3 |
| Molecular Weight | 357.49 |
| CAS Registry Number | 2547-66-2 |
| EC Number | 219-831-7 |
| SMILES | C1N(CN(CN1CC2=CC=CC=C2)CC3=CC=CC=C3)CC4=CC=CC=C4 |
| Density | 1.1±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 49 - 51 °C (Expl.) |
| Boiling point | 465.1±45.0 °C 760 mmHg (Calc.)*, 502.8 °C (Expl.) |
| Flash point | 234.5±19.3 °C (Calc.)*, 113 °C (Expl.) |
| Index of refraction | 1.631 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
1,3,5-Tribenzyl-1,3,5-triazinane is a saturated heterocyclic compound consisting of a six-membered 1,3,5-triazinane ring with nitrogen atoms at positions 1, 3, and 5, each substituted with a benzyl group. Its molecular formula is C27H27N3, and it has a molecular weight of approximately 393.53 g/mol. The triazinane ring is fully saturated, giving the molecule a flexible three-dimensional structure, while the three benzyl substituents introduce steric bulk and hydrophobic character that influence solubility, crystallization, and reactivity. The compound is typically synthesized via condensation of formaldehyde with benzylamine in a 1:3 stoichiometric ratio under controlled conditions. This Mannich-type reaction results in cyclization to form the 1,3,5-triazinane ring while installing the three benzyl groups on the nitrogen atoms. Reaction conditions such as temperature, solvent, and pH are carefully optimized to promote selective cyclization and to minimize polymerization or formation of oligomeric byproducts. Purification is usually achieved by recrystallization or chromatography to isolate the pure triazinane derivative. Chemically, 1,3,5-tribenzyl-1,3,5-triazinane is relatively stable under ambient conditions. The nitrogen atoms in the ring are tertiary due to benzyl substitution, which limits their basicity and reactivity toward nucleophilic or electrophilic attack. The benzyl groups, however, provide reactive sites for oxidative cleavage or hydrogenolysis if further transformation is desired. The saturated ring system allows for flexibility, and the nitrogen atoms can coordinate with metal centers, making the compound of interest in coordination chemistry. The compound is generally a solid at room temperature with good solubility in nonpolar and moderately polar organic solvents such as chloroform, dichloromethane, or ethanol. The steric hindrance provided by the benzyl substituents influences both crystal packing and solubility, and it can affect the reactivity in reactions where steric accessibility of the nitrogen atoms is important. In practical applications, 1,3,5-tribenzyl-1,3,5-triazinane serves as an intermediate or protective agent in organic synthesis. The benzyl groups can act as temporary protecting groups for the nitrogen atoms, allowing selective functionalization of other sites in multistep synthetic sequences. After the desired transformations, the benzyl groups can be removed under reductive or hydrogenolytic conditions to regenerate the free triazinane core. The molecule also finds utility in studies of nitrogen-containing heterocycles due to its defined geometry, symmetry, and the possibility for further derivatization. Overall, the combination of a saturated nitrogen-containing ring with sterically bulky benzyl substituents provides 1,3,5-tribenzyl-1,3,5-triazinane with unique structural features. Its stability, solubility, and potential for selective transformation make it a valuable building block in organic synthesis and heterocyclic chemistry, enabling the design of more complex molecules for research and application in chemical and material sciences. References 2021. Coordination of Hexahydro-1,3,5-trimethyl-1,3,5-triazine in Cadmium(II) and Cobalt(II) Carboxylate Complexes. Russian Journal of Coordination Chemistry, 47(8). DOI: 10.1134/s1070328421080078 2018. Facile Access to 3-Unsubstituted Tetrahydroisoquinolonic Acids via the Castagnoli–Cushman Reaction. Synthesis. DOI: 10.1055/s-0036-1591923 |
| Market Analysis Reports |