Isopropylaminosulfonyl chloride (C3H8ClNO2S) is an organosulfur compound containing a sulfonyl chloride (-SO2Cl) functional group attached to an isopropylamine (-NHCH(CH3)2) moiety. Sulfonyl chlorides are widely recognized for their role as reactive intermediates in organic synthesis, particularly in the preparation of sulfonamides, sulfonylureas, and other sulfur-containing compounds.
The synthesis of isopropylaminosulfonyl chloride typically involves the reaction of isopropylamine with chlorosulfonic acid (HSO3Cl) or similar chlorinating agents. This reaction results in the introduction of the sulfonyl chloride group, making the compound a valuable intermediate for further chemical modifications. Careful control of reaction conditions is necessary to minimize byproduct formation and ensure high yields.
In organic synthesis, isopropylaminosulfonyl chloride serves as a key reagent for the introduction of the sulfonyl functional group in pharmaceutical and agrochemical compounds. Sulfonamides derived from this compound are of particular interest due to their applications in medicinal chemistry. Sulfonyl-containing molecules are often found in biologically active substances, including antibiotics, diuretics, and enzyme inhibitors. The presence of the isopropyl group can influence the solubility, stability, and biological activity of the resulting derivatives.
Isopropylaminosulfonyl chloride also plays a role in material science, particularly in the modification of polymeric structures. The incorporation of sulfonyl groups into polymers can improve their chemical resistance, mechanical properties, and thermal stability. Functionalized polymers containing sulfonyl groups are used in various industrial applications, including ion-exchange membranes and specialty coatings.
Handling of isopropylaminosulfonyl chloride requires appropriate safety measures due to its reactivity. Sulfonyl chlorides are known to be moisture-sensitive and can react with water to form acidic byproducts. Proper storage conditions, such as low-temperature environments and sealed containers, are essential to maintain the stability of the compound. Personal protective equipment and ventilation are necessary to prevent exposure, as sulfonyl chlorides can cause irritation to the skin, eyes, and respiratory system.
Research into sulfonyl chlorides, including isopropylaminosulfonyl chloride, continues to expand their applications in chemical synthesis and material development. The study of these compounds contributes to advancements in pharmaceuticals, agricultural chemicals, and functional materials, reinforcing their significance in industrial and academic research.
References
none
|













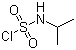

 GHS05;GHS07 Danger Details
GHS05;GHS07 Danger Details