Online Database of Chemicals from Around the World
| UnaveraChemLab GmbH | Germany | |||
|---|---|---|---|---|
 | www.unavera.de | |||
 | +49 (0) 8823-1351 | |||
 | +49 (0) 8823-3449 | |||
 | axel@unavera.de | |||
| Chemical manufacturer since 1982 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hebei Winjoy United Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.winjoy-uni.com | |||
 | +86 17734561860 | |||
 | winjoy@winjoy-uni.com | |||
 | QQ Chat | |||
 | WeChat: 17734561860 | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Aaron Chemistry GmbH | Germany | |||
|---|---|---|---|---|
 | www.aaron-chemistry.de | |||
 | +49 (8823) 917-521 | |||
 | +49 (8823) 917-523 | |||
 | sales@aaron-chemistry.de | |||
| Chemical manufacturer | ||||
| Beijing Bayi Space LCD Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bayi.com.cn | |||
 | +86 (10) 6976-5588 ex 8068 +86 18600516188 | |||
 | +86 (10) 6976-0560 | |||
 | zhangshupo@aliyun.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2004 | ||||
| Yantai Winning Fine Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.winningchem.com | |||
 | +86 (535) 557-0816 | |||
 | +86 (535) 557-3078 | |||
 | sales@winningchem.com | |||
| Chemical manufacturer | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Alcohol |
|---|---|
| Name | 2-Butylpropane-1,3-diol |
| Synonyms | 2-n-Butylpropane-1,3-diol |
| Molecular Structure | 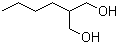 |
| Molecular Formula | C7H16O2 |
| Molecular Weight | 132.20 |
| CAS Registry Number | 2612-26-2 |
| EC Number | 677-792-5 |
| SMILES | CCCCC(CO)CO |
| Density | 0.9±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.450, Calc.* |
| Boiling Point | 242.8±8.0 °C (760 mmHg), Calc.* |
| Flash Point | 112.3±13.0 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS08 Warning Details GHS07;GHS08 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H317-H361 Details | ||||||||||||
| Safety Statements | P203-P261-P264-P270-P272-P280-P301+P317-P302+P352-P318-P321-P330-P333+P317-P362+P364-P405-P501 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
2-Butylpropane-1,3-diol is an organic compound that belongs to the family of diols, which are characterized by the presence of two hydroxyl groups (-OH) attached to adjacent or non-adjacent carbon atoms. This compound consists of a butyl group (C4H9) attached to the second carbon of a propane-1,3-diol backbone. The chemical structure of 2-butylpropane-1,3-diol gives it both hydrophobic and hydrophilic properties, which make it a versatile compound for a range of applications in the chemical and material industries. The discovery of 2-butylpropane-1,3-diol is associated with the broader search for functionalized diols that could serve as intermediates in the synthesis of various industrial chemicals, including biodegradable polymers, surfactants, and solvents. Its structure, which combines the properties of both an alcohol and a butyl group, makes it suitable for use in the development of products that require a balance between water solubility and oil solubility. Synthesis of 2-butylpropane-1,3-diol is typically achieved through the reaction of propylene oxide with butyl groups in the presence of a catalyst. This process allows for the introduction of the butyl group onto the second carbon atom of the propane backbone, creating a diol that can be further functionalized or incorporated into polymer systems. The reaction conditions are relatively mild, making this method an attractive option for large-scale production. One of the primary applications of 2-butylpropane-1,3-diol is in the production of polyesters and polyurethanes, where it is used as a monomer to provide flexibility, hydrophobicity, and improved processing characteristics. In these polymers, the butyl group imparts water resistance and helps to enhance the stability of the final product under different environmental conditions. 2-Butylpropane-1,3-diol can also be used as a modifier in existing polymer formulations to improve the mechanical properties and overall performance of coatings, adhesives, and sealants. In addition to its use in the polymer industry, 2-butylpropane-1,3-diol has found applications in the manufacture of lubricants and surfactants. The combination of the butyl group and hydroxyl groups provides an excellent balance of solubility and lubricity, making it useful in formulations that require both water and oil compatibility. Its mild chemical reactivity also makes it useful as a stabilizing agent in various chemical reactions, especially in the preparation of emulsions or other dispersed systems. Moreover, the compound has been studied for potential use in pharmaceutical formulations and cosmetic products. Due to its relatively low toxicity and ability to interact gently with other ingredients, it has been proposed as a safe ingredient in skin care products and personal care items. Its presence can enhance the texture and stability of emulsions, while also providing mild humectant properties. The versatility of 2-butylpropane-1,3-diol continues to be explored in various industrial and research applications. As manufacturers and researchers aim to develop more sustainable materials, this compound’s ability to bridge the gap between hydrophobic and hydrophilic environments makes it a valuable tool in the creation of advanced materials and chemicals. References 2012. Promising directions for utilization of glycerol-containing waste from biodiesel fuel production. Russian Journal of General Chemistry. DOI: 10.1134/s1070363212050362 2007. Microbial asymmetric oxidation of 2-butyl-1,3-propanediol. Applied Microbiology and Biotechnology. DOI: 10.1007/s00253-007-0974-0 2008. Synthesis of 3-alkyl(aryl)thietanes. Petroleum Chemistry. DOI: 10.1134/s0965544108020084 |
| Market Analysis Reports |