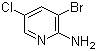
2-Amino-3-bromo-5-chloropyridine is a noteworthy chemical compound with valuable applications in organic synthesis and medicinal chemistry. This heterocyclic compound contains an amino group, a bromine atom, and a chlorine atom attached to a pyridine ring, making it a versatile intermediate for various chemical transformations.
The discovery of 2-Amino-3-bromo-5-chloropyridine is rooted in efforts to develop new pyridine derivatives with specific electronic and steric properties. Pyridine, a six-membered aromatic ring containing one nitrogen atom, is a key structure in many bioactive molecules and ligands. The introduction of bromine and chlorine substituents at specific positions on the pyridine ring provides additional functionality and reactivity, enhancing the compound's utility in various chemical processes.
The synthesis of 2-Amino-3-bromo-5-chloropyridine typically involves a multi-step process starting from commercially available pyridine derivatives. The synthesis begins with the introduction of the amino group, which can be achieved by nitration or other nitrogen-insertion methods. Following this, selective bromination and chlorination are performed to introduce the bromine and chlorine atoms at the 3- and 5-positions, respectively. The reaction conditions are optimized to ensure high selectivity and yield of the desired product, and the compound is purified using techniques such as recrystallization or chromatography.
One of the primary applications of 2-Amino-3-bromo-5-chloropyridine is in organic synthesis, where it serves as a building block for the preparation of more complex molecules. The presence of both bromine and chlorine substituents makes it a useful intermediate for cross-coupling reactions, where these halogen atoms can participate in various chemical transformations. For example, 2-Amino-3-bromo-5-chloropyridine can be used in Suzuki-Miyaura or Sonogashira coupling reactions to form carbon-carbon bonds, facilitating the synthesis of diverse organic compounds.
In medicinal chemistry, 2-Amino-3-bromo-5-chloropyridine is employed as a precursor in the development of pharmaceuticals. The unique combination of functional groups provides opportunities for modifying biological activity and optimizing drug-like properties. The compound can be used to synthesize various heterocyclic derivatives that have potential applications as drugs or therapeutic agents. For instance, it can be used to prepare pyridine-based compounds with anti-inflammatory, anti-cancer, or antimicrobial properties.
In addition to its use in organic synthesis and medicinal chemistry, 2-Amino-3-bromo-5-chloropyridine has applications in materials science. The compound can be employed in the development of new materials with specific electronic or optical properties. Its ability to participate in coordination chemistry allows for the creation of novel metal complexes that may have applications in catalysis or sensors.
The advantages of using 2-Amino-3-bromo-5-chloropyridine include its versatility as a chemical intermediate and its ability to participate in a range of reactions due to its halogen substituents. However, challenges may include managing the reactivity of the halogen groups and optimizing reaction conditions to achieve the desired products.
Future research on 2-Amino-3-bromo-5-chloropyridine may focus on exploring new applications in synthetic chemistry, optimizing its use in pharmaceutical development, and investigating its potential in materials science. Researchers may also explore modifications to the compound's structure to enhance its properties and expand its utility in various chemical processes.
References
2004. Palladium-Catalyzed Cross-Coupling Reactions in the Synthesis of Pharmaceuticals. Organometallics in Process Chemistry.
DOI: 10.1007/b94551
1971. Reaction of 2-amino-5-halopyridines with methyl acrylate. Chemistry of Heterocyclic Compounds.
DOI: 10.1007/bf00473087
|











































































 GHS05;GHS07 Danger Details
GHS05;GHS07 Danger Details