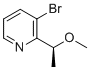
(S)-3-Bromo-2-(1-methoxyethyl)pyridine is a chiral brominated pyridine derivative used in medicinal chemistry as a versatile intermediate for the preparation of complex bioactive molecules. It contains a pyridine ring substituted at the 2-position with an (S)-configured 1-methoxyethyl group and a bromine atom at the 3-position, providing both chirality and a reactive handle for further derivatization. The stereogenic center adjacent to the heteroaromatic ring enables stereocontrolled construction of chiral scaffolds relevant to pharmaceutical development, particularly in programs targeting kinase modulation, inflammatory pathways, and central nervous system applications.
The bromine substituent serves as a selective activation point for palladium-catalyzed cross-coupling reactions such as Suzuki–Miyaura, Sonogashira, and Buchwald–Hartwig processes, facilitating the introduction of aryl, heteroaryl, alkynyl, or amine fragments. Meanwhile, the methoxyethyl side chain may be elaborated by controlled deprotection or functional group modification, supporting modular diversification. This combination of a halogenated heteroaromatic ring and a chiral alkoxy substituent gives the compound a favorable balance of reactivity, stereochemical information, and structural rigidity, making it suitable for incorporation into small-molecule frameworks with defined three-dimensional orientation.
Patent literature increasingly documents the use of this intermediate in multi-step syntheses of advanced therapeutic candidates. It most often appears as a protected chiral building block introduced early in synthetic sequences, ensuring preservation of stereochemical purity throughout downstream functionalization. The compound’s enantiomeric enrichment and purity are typically monitored by chiral HPLC and optical rotation methods in industrial settings. Its utility aligns with current medicinal chemistry strategies emphasizing heteroaryl motifs, precise stereochemistry, and compatibility with metal-catalyzed transformations in scalable synthetic schemes.
|




 GHS07 Warning Details
GHS07 Warning Details