Online Database of Chemicals from Around the World
| F&F Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fnfchem.com | |||
 | +86 (10) 6444-6910 | |||
 | +86 (10) 6444-6915 | |||
 | heyifnfchem@163.com heyi@fnfchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Nile Chemicals | India | |||
|---|---|---|---|---|
 | www.nilechemicals.com | |||
 | +91 (22) 6631-3162 | |||
 | +91 (22) 2205-4833 | |||
 | sales@nilechemicals.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Watec Laboratories, Inc. | China | |||
|---|---|---|---|---|
 | www.wateclaboratories.com | |||
 | +86 (519) 8692-1516 +86 18602586511 | |||
 | +86 (519) 8526-7382 | |||
 | info@wateclaboratories.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2012 | ||||
| LOBA Feinchemie AG | Austria | |||
|---|---|---|---|---|
 | www.loba.co.at | |||
 | +43 (223) 277-391 | |||
 | +43 (223) 276-677 | |||
 | sales@loba.co.at | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2012 | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridine compound |
|---|---|
| Name | Ferrozine disodium salt |
| Synonyms | Disodium 4,4'-[3-(pyridin-2-yl)-1,2,4-triazine-5,6-diyl]bis(benzenesulphonate); 3-(2-Pyridyl)-5,6-bis(4-sulfophenyl)-1,2,4-triazine disodium salt |
| Molecular Structure | 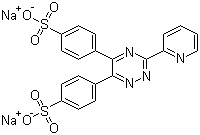 |
| Molecular Formula | C20H12N4Na2O6S2 |
| Molecular Weight | 514.44 |
| CAS Registry Number | 28048-33-1 |
| EC Number | 248-797-6 |
| SMILES | C1=CC=NC(=C1)C2=NC(=C(N=N2)C3=CC=C(C=C3)S(=O)(=O)[O-])C4=CC=C(C=C4)S(=O)(=O)[O-].[Na+].[Na+] |
| Melting point | >300 °C |
|---|---|
| Water solubility | soluble |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Ferrazine disodium salt is a water-soluble compound and a highly specific reagent for iron quantification. Its systematic name is 4,4'-(3-phenyl-1,2,4-triazine-5,6-diyl)bis(benzenesulfonic acid) disodium salt. The discovery of ferrazine is of vital importance to analytical chemistry because of its ability to form a stable and brightly colored complex with iron, making it an essential tool for iron determination in various fields. Ferrazine was discovered in the mid-20th century, when efforts were underway to develop more accurate and efficient methods for detecting and measuring metal ions in solution. Prior to the introduction of ferrazine, existing iron detection methods were often interfered with by other metal ions and lacked the sensitivity required for accurate quantification at low concentrations. The development of ferrazine addressed these issues, providing a reagent with high specificity for iron (II) ions. The main application of ferrazine disodium salt is the colorimetric determination of iron. When ferrazine reacts with iron (II) ions, a magenta complex is formed. This complex exhibits strong absorbance at 562 nm, allowing for accurate spectrophotometric measurements. The intensity of the color produced is proportional to the iron concentration in the sample, allowing for accurate quantification. This property makes ferrazine an indispensable reagent in various fields including environmental science, biochemistry, and clinical diagnostics. In environmental science, ferrazine disodium salt is used to measure iron levels in water, soil, and other environmental samples. Accurate iron quantification is essential for monitoring and controlling pollution, as iron plays an important role in various biochemical and geochemical processes. In biochemistry and molecular biology, ferrazine disodium salt is used to assess iron levels in biological samples such as blood, serum, and tissues. Iron is an essential element for many biological processes, including oxygen transport, DNA synthesis, and electron transport in mitochondria. Abnormal iron levels can indicate the presence of various health conditions, including anemia, hemochromatosis, and iron deficiency. Clinical laboratories use ferrazine-based assays to diagnose and monitor these conditions, providing valuable information for patient care. Additionally, ferrazine is used to study iron metabolism and homeostasis. Iron is an essential nutrient, but its levels must be tightly controlled to prevent toxicity. Researchers use ferrazine to study the mechanisms of iron absorption, storage, and regulation in cells and organisms. This could help understand diseases associated with iron metabolism and develop potential treatments. In addition to its application in aqueous solutions, ferrazine can also be used in solid phase assays. Immobilizing ferrazine on a solid support allows the development of sensors and test strips for rapid and on-site iron detection. This extends the use of ferrazine beyond the laboratory setting, enabling its application in the field, such as monitoring iron levels in agricultural soil or industrial wastewater. The use of ferrazine disodium salt is relatively simple, requiring only the preparation of a reagent solution that is mixed with the sample to be analyzed. The formation of the colored complex is rapid and stable, allowing for fast and reliable measurements. Furthermore, the high specificity of ferrazine for iron (II) minimizes interference from other metal ions, although it is often used with a reducing agent to ensure that all iron in the sample is in the iron (II) state. References 2024. Iron overload promotes hemochromatosis-associated osteoarthritis via the mTORC1-p70S6K/4E-BP1 pathway. Arthritis Research & Therapy, 26(1). DOI: 10.1186/s13075-024-03276-9 2021. Complexation of ferrous ions by ferrozine, 2,2′-bipyridine and 1,10-phenanthroline: Implication for the quantification of iron in biological systems. Journal of Inorganic Biochemistry, 223. DOI: 10.1016/j.jinorgbio.2021.111546 2019. Microwave-Assisted Extraction Combined with In-Capillary [Fe(ferrozine)3]2+-CE-DAD to Screen Active Components with the Ability to Chelate Ferrous Ions from Flos Sophorae Immaturus (Flos Sophorae). Molecules, 24(15). DOI: 10.3390/molecules24152710 |
| Market Analysis Reports |