Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Yantai Liquid Crystal Materials Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ytderun.com | |||
 | +86 (535) 630-0169 | |||
 | +86 (535) 630-0169 | |||
 | yangli@ytderun.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Shanghai Yishenghui Bio-medical Technologies Inc. | China | |||
|---|---|---|---|---|
 | www.ychemical.com | |||
 | +86 (21) 6145-3981 +86 13564731332 | |||
 | +86 (21) 6145-3981 | |||
 | yishenghui99@126.com | |||
 | QQ Chat | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Zhangjiagang Clent Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.clentchem.com | |||
 | +86 (512) 5881-3106 +86 13962460297 | |||
 | +86 (512) 5881-2106 | |||
 | sales@clentchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Shanghai Edu Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemido.com | |||
 | +86 19301251526 | |||
 | sales1@chemido.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Molekula Ltd | UK | |||
|---|---|---|---|---|
 | www.molekula.com | |||
 | +44 (1747) 831-066 | |||
 | +44 (1747) 831-199 | |||
 | info@molekula.com | |||
| Chemical manufacturer | ||||
| Ricci Chimica | Italy | |||
|---|---|---|---|---|
 | www.riccichimica.com | |||
 | +39 (75) 692-9241 | |||
 | +39 (75) 592-6595 | |||
 | info@riccichimica.com | |||
| Chemical manufacturer | ||||
| AOBChem USA | USA | |||
|---|---|---|---|---|
 | www.aobchem.com | |||
 | +1 (323) 382-7678 | |||
 | +1 (213) 687-8698 | |||
 | sales@aobchem.com | |||
| Chemical manufacturer | ||||
| Xinyi Chemical Research Institute | China | |||
|---|---|---|---|---|
 | www.xinyichemical.com | |||
 | +86 (519) 8646-2478 8647-1022 | |||
 | +86 (519) 8646-8478 | |||
 | lxy.mq@163.com | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Organic fluorine compound >> Fluorophenol series |
|---|---|
| Name | 2,6-Difluorophenol |
| Molecular Structure | 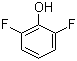 |
| Molecular Formula | C6H4F2O |
| Molecular Weight | 130.09 |
| CAS Registry Number | 28177-48-2 |
| EC Number | 248-884-9 |
| SMILES | C1=CC(=C(C(=C1)F)O)F |
| Density | 1.4±0.1 g/cm3 Calc.*, 1.27 g/mL (Expl.) |
|---|---|
| Melting point | 38 - 41 °C (Expl.) |
| Boiling point | 157.7 °C 760 mmHg (Calc.)*, 179 - 181.7 °C (Expl.) |
| Flash point | 58.9 °C (Calc.)*, 58 °C (Expl.) |
| Index of refraction | 1.496 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS02;GHS05;GHS07 Danger Details GHS02;GHS05;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H228-H302-H312-H314-H315-H319-H335 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P240-P241-P260-P261-P264-P264+P265-P270-P271-P280-P301+P317-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P351+P338-P305+P354+P338-P316-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P363-P370+P378-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 1325 | ||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||
|
2,6-Difluorophenol is an aromatic compound in which a phenol ring is substituted with fluorine atoms at the 2 and 6 positions relative to the hydroxyl group. Its molecular formula is C6H4F2O, with a molecular weight of approximately 126.10 g/mol. The presence of electronegative fluorine atoms at the ortho positions to the hydroxyl group significantly influences the acidity, electronic distribution, and reactivity of the molecule. The hydroxyl group enables hydrogen bonding, contributing to solubility in polar solvents and providing a reactive site for esterification, etherification, or metal coordination. Synthesis of 2,6-difluorophenol typically involves selective fluorination of phenol derivatives using electrophilic or nucleophilic fluorination strategies. One approach is the direct fluorination of phenol under controlled conditions to favor substitution at the 2 and 6 positions. Alternatively, the compound can be obtained via the hydrolysis of 2,6-difluorophenyl derivatives, such as 2,6-difluorophenyl halides or nitro compounds, under appropriate reaction conditions. Regioselectivity is controlled by the directing effects of the hydroxyl group and by steric hindrance from existing substituents. Chemically, 2,6-difluorophenol exhibits higher acidity compared to unsubstituted phenol due to the electron-withdrawing effect of the ortho-fluorine atoms, which stabilize the phenoxide ion upon deprotonation. The hydroxyl proton can participate in acid–base reactions, forming salts with bases, or undergo esterification with acyl chlorides and anhydrides. The fluorine substituents reduce the reactivity of the aromatic ring toward electrophilic substitution at the ortho and para positions, while slightly activating the meta positions relative to the hydroxyl group. The compound is typically a solid at room temperature and exhibits moderate solubility in polar organic solvents such as ethanol, acetone, and dimethylformamide. Its chemical stability is generally good under ambient conditions, although strong bases or nucleophiles can react with the phenolic hydroxyl group or induce substitution of the fluorine atoms under harsh conditions. The presence of two ortho fluorine atoms also introduces steric hindrance that can influence the molecule’s interactions in both chemical and biological systems. In practical applications, 2,6-difluorophenol is commonly used as a building block in organic synthesis, pharmaceuticals, and agrochemical development. The molecule’s hydroxyl group allows derivatization, while the fluorine atoms can modify electronic properties, lipophilicity, and metabolic stability in bioactive compounds. It serves as a precursor for the preparation of ethers, esters, and other functionalized derivatives, enabling the design of molecules with targeted chemical and biological properties. The combination of a reactive hydroxyl group and sterically hindered fluorine-substituted aromatic ring makes 2,6-difluorophenol a valuable intermediate for synthetic chemistry. Its defined electronic and steric characteristics support controlled functionalization and selective reactivity, allowing it to be incorporated into more complex molecular frameworks for research, development, and applied chemical purposes. References 2010. Modulation of the Aerobic Oxidative Polymerization in Phenylazomethine Dendrimers Assembling Copper Complexes. Chemistry - A European Journal. DOI: 10.1002/chem.201001516 2010. A Diverse Series of Substituted Benzenesulfonamides as Aldose Reductase Inhibitors with Antioxidant Activity: Design, Synthesis, and in Vitro Activity. Journal of Medicinal Chemistry. DOI: 10.1021/jm101008m 2004. Degradation of 2-fluorophenol by the brown-rot fungus Gloeophyllum striatum : evidence for the involvement of extracellular Fenton chemistry. Applied Microbiology and Biotechnology. DOI: 10.1007/s00253-003-1445-x |
| Market Analysis Reports |