Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Shanghai Rory Fine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rorychem.com | |||
 | +86 (21) 5070-2305 | |||
 | +86 (21) 5070-4994 | |||
 | rorychem@hotmail.com 13701863355@139.com | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Futoh Chemicals Co., Ltd. | Japan | |||
|---|---|---|---|---|
 | www.futoh.co.jp | |||
 | +81 (52) 521-8171 | |||
 | +81 (52) 521-8793 | |||
 | tokoro@futoh.co.jp | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Changzhou Sunchem Pharmaceutical Chemical Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sunkechem.com | |||
 | +86 (519) 8519-5575 8519-5528 8519-5565 | |||
 | +86 (519) 8519-5585 | |||
 | info@sunkechem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Qingdao Kingway Pharmtech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingwaypharm.com | |||
 | +86 (532) 8711-8899 | |||
 | +86 (532) 8411-7788 | |||
 | sales@kingwaypharm.com kingwaypharm@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Hangzhou Loop Biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.looppharm.com | |||
 | +86 (0571) 8526-0761 | |||
 | sales01@looppharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Hangzhou Zentra Bio-Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zentrachem.com | |||
 | 18321999949 | |||
 | steven@zentrachem.com | |||
 | QQ Chat | |||
 | WeChat: Hangzhou Zentra Bio-Chemical Co., Ltd. | |||
| Chemical distributor since 2025 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Jiangyin Trust-Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.trust-chem.com | |||
 | +86 (510) 8625-7670 8626-8020 8626-9706 | |||
 | +86 (510) 8626-8021 | |||
 | trust@hi2000.com | |||
| Chemical manufacturer since 1996 | ||||
| Classification | Chemical reagent >> Organic reagent >> Fatty ketone (including enol) |
|---|---|
| Name | 5-Hydroxy-1-tetralone |
| Synonyms | 1,2,3,4-Tetrahydro-5-hydroxynaphthalen-1-one |
| Molecular Structure | 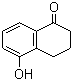 |
| Molecular Formula | C10H10O2 |
| Molecular Weight | 162.19 |
| CAS Registry Number | 28315-93-7 |
| EC Number | 248-958-0 |
| SMILES | C1CC2=C(C=CC=C2O)C(=O)C1 |
| Density | 1.2±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 206 - 209 °C (Expl.) |
| Boiling point | 325.3±31.0 °C 760 mmHg (Calc.)* |
| Flash point | 138.1±17.4 °C (Calc.)* |
| Index of refraction | 1.603 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
5-Hydroxy-1-tetralone is an organic compound belonging to the class of hydroxy-substituted tetralones. Structurally, it consists of a tetralone core—a bicyclic system combining a benzene ring fused to a cyclohexanone ring—with a hydroxyl (–OH) group attached at the 5-position of the aromatic ring. The ketone functionality is located at the 1-position of the cyclohexanone ring, which is adjacent to the aromatic ring fusion. The tetralone framework serves as an important scaffold in organic synthesis due to its rigid bicyclic structure and the presence of both aromatic and ketone functional groups. Hydroxy substitution at the 5-position modifies the electronic and steric environment of the molecule, often influencing reactivity and potential biological activity. Such substitution can facilitate further functionalization through typical reactions of phenolic groups, including electrophilic aromatic substitution, esterification, and etherification. Synthesis of 5-hydroxy-1-tetralone can be accomplished by various methods. One common approach involves hydroxylation of 1-tetralone derivatives, typically using selective oxidizing agents or electrophilic substitution reactions directed to the 5-position on the aromatic ring. Alternative routes may include cyclization strategies where the hydroxy group is introduced during ring formation. The synthesis aims to achieve regioselectivity to ensure hydroxylation specifically at the 5-position. Chemically, 5-hydroxy-1-tetralone exhibits properties characteristic of both phenols and cyclic ketones. The phenolic hydroxyl group can act as a weak acid, donating hydrogen in hydrogen bonding and participating in redox reactions. The ketone group at the 1-position undergoes nucleophilic addition and enolization, allowing participation in condensation and reduction reactions. The combination of these functional groups in a bicyclic structure offers versatility for synthetic modifications. Applications of 5-hydroxy-1-tetralone include its use as an intermediate in organic synthesis and medicinal chemistry. It serves as a building block for the preparation of more complex molecules, including natural product analogues, pharmaceuticals, and functional materials. Its phenolic and ketone functionalities enable derivatization into compounds with diverse chemical and biological properties. Analytical characterization of 5-hydroxy-1-tetralone typically involves spectroscopic techniques. Proton nuclear magnetic resonance (²H NMR) spectroscopy identifies aromatic protons, hydroxyl proton signals, and methylene protons adjacent to the ketone. Carbon-13 NMR spectroscopy confirms the presence of ketone carbonyl and aromatic carbons. Infrared (IR) spectroscopy detects characteristic absorption bands for hydroxyl groups (around 3400 cm−1) and ketone carbonyl stretch (near 1700 cm−1). Mass spectrometry provides molecular weight confirmation and fragmentation patterns consistent with the compound’s structure. Physically, 5-hydroxy-1-tetralone is generally a solid at room temperature, often crystalline in form. It exhibits moderate solubility in organic solvents such as ethanol, methanol, and dichloromethane, while its solubility in water is limited due to its largely hydrophobic bicyclic structure. In summary, 5-hydroxy-1-tetralone is a hydroxy-substituted bicyclic ketone combining phenolic and ketone functionalities within a tetralone framework. Its chemical properties and structural features make it a valuable intermediate in synthetic organic chemistry and a potential precursor for the development of biologically active compounds. References 1972. Fluorometric determination of glucose in cerebrospinal fluid and blood by the revised 5-hydroxy-1-tetralone method. Biochemical Medicine, 6(2). DOI: 10.1016/0006-2944(72)90026-9 1966. Eine Verbesserung der fluorimetrischen Blutglucosebestimmung mit 5-Hydroxytetralon. Fresenius' Zeitschrift für analytische Chemie. DOI: 10.1007/bf00521767 |
| Market Analysis Reports |