Online Database of Chemicals from Around the World
| Biosynth AG. | Switzerland | |||
|---|---|---|---|---|
 | www.biosynth.com | |||
 | +41 (71) 858-2020 | |||
 | +41 (71) 858-2030 | |||
 | welcome@biosynth.ch | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Analytical chemistry >> Standard >> Pharmacopoeia standards and magazine standards |
|---|---|
| Name | 3-Nitro-4-phenoxy-5-sulfamoylbenzoic acid |
| Molecular Structure | 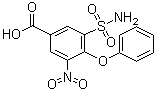 |
| Molecular Formula | C13H10N2O7S |
| Molecular Weight | 338.29 |
| CAS Registry Number | 28328-53-2 |
| EC Number | 248-970-6 |
| SMILES | C1=CC=C(C=C1)OC2=C(C=C(C=C2S(=O)(=O)N)C(=O)O)[N+](=O)[O-] |
| Solubility | Practically insoluble (0.024 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.585±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2014 ACD/Labs) |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
3-Nitro-4-phenoxy-5-sulfamoylbenzoic acid is a significant chemical compound with notable applications in various fields, including medicinal chemistry and environmental science. This compound is recognized for its unique structural features and its utility in research and practical applications. The discovery of 3-nitro-4-phenoxy-5-sulfamoylbenzoic acid arose from efforts to develop new chemical entities with specific properties. The compound is characterized by its benzoic acid core, which is substituted with a nitro group, a phenoxy group, and a sulfamoyl group. These substituents impart distinctive chemical and biological properties to the molecule, making it a subject of interest in several scientific domains. The synthesis of 3-nitro-4-phenoxy-5-sulfamoylbenzoic acid involves several chemical reactions that introduce each functional group to the benzoic acid backbone. Typically, the process starts with the nitration of benzoic acid to introduce the nitro group. Subsequent reactions involve the introduction of the phenoxy group and the sulfamoyl group through various chemical transformations, such as nucleophilic substitution and sulfonation. These synthetic steps are designed to ensure the correct positioning and functionalization of the substituents. In medicinal chemistry, 3-nitro-4-phenoxy-5-sulfamoylbenzoic acid has been explored for its potential therapeutic applications. The compound's ability to interact with biological targets, such as enzymes or receptors, makes it a candidate for drug development. For instance, the sulfamoyl group is known for its role in drug design due to its potential to interact with biological systems, while the nitro and phenoxy groups contribute to the molecule's overall activity and selectivity. Research into these interactions helps to identify the compound's efficacy and safety profile for potential medical use. Beyond medicinal chemistry, 3-nitro-4-phenoxy-5-sulfamoylbenzoic acid finds applications in environmental science. Its chemical properties make it useful in the study of pollutants and their effects on ecosystems. The compound's ability to form complexes with metals or interact with other environmental substances provides insights into its environmental behavior and potential impact. This information is crucial for developing methods to mitigate pollution and manage chemical contaminants in various settings. In summary, 3-nitro-4-phenoxy-5-sulfamoylbenzoic acid is a chemically complex compound with diverse applications in medicinal chemistry and environmental science. Its unique structure and functional groups contribute to its utility in research and practical applications, making it a valuable subject of study. References 2003. Piretanide. Pharmaceutical Substances. https://pharmaceutical-substances.thieme.com/ps/search-results?docUri=KD-16-0135 2003. Bumetanide. Pharmaceutical Substances. https://pharmaceutical-substances.thieme.com/ps/search-results?docUri=KD-02-0139 List of 7074 potential endocrine disrupting compounds (EDCs) by PARC T4.2. DOI: 10.5281/zenodo.10944198 |
| Market Analysis Reports |