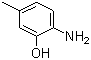
6-Amino-m-cresol, also known as 6-amino-3-methylphenol, is an aromatic compound with the molecular formula C7H9NO. Its structure consists of a benzene ring substituted with an amino group (–NH2) at the 6-position, a hydroxyl group (–OH) at the 1-position, and a methyl group (–CH3) at the 3-position relative to the hydroxyl group. This arrangement classifies it as a derivative of m-cresol (3-methylphenol) featuring an additional amino substituent.
The synthesis of 6-amino-m-cresol typically involves nitration of m-cresol followed by selective reduction of the nitro group to the amino group. Controlling reaction conditions is crucial to achieve the desired substitution pattern without producing unwanted isomers or over-reduction. Alternative methods include amination reactions on suitably functionalized aromatic precursors. Purification and characterization techniques such as crystallization, chromatography, nuclear magnetic resonance (NMR) spectroscopy, and infrared (IR) spectroscopy are commonly employed to confirm the compound’s identity and purity.
6-Amino-m-cresol is predominantly utilized in the cosmetic industry as an intermediate in the manufacture of oxidative hair dyes. Its chemical structure—with both amino and hydroxyl groups—facilitates participation in oxidative coupling reactions with other dye precursors in the presence of oxidizing agents such as hydrogen peroxide. These reactions result in the formation of chromophores that impart various shades and tones to hair, commonly producing colors in the brown to black spectrum. The methyl group influences the electronic properties and steric factors of the molecule, which can affect dyeing performance and shade development.
Beyond its role in hair dye formulations, 6-amino-m-cresol serves as a versatile building block in organic synthesis. The functional groups present in the molecule allow it to engage in a wide array of chemical reactions, including electrophilic substitution, diazotization, and condensation reactions. Diazotization of the amino group forms diazonium salts, which are key intermediates in the synthesis of azo dyes and other pigment compounds. The hydroxyl group can participate in etherification, esterification, and hydrogen bonding, affecting solubility and reactivity.
The compound’s physicochemical properties include moderate solubility in water and various polar organic solvents. Analytical techniques for its characterization include NMR spectroscopy, which provides detailed information on proton and carbon environments; IR spectroscopy, highlighting characteristic absorptions of amino and hydroxyl groups; and chromatographic methods such as high-performance liquid chromatography (HPLC), which are used to assess purity and detect impurities.
Safety considerations for 6-amino-m-cresol are aligned with those for many aromatic amines and phenols. It may cause skin and eye irritation and has the potential to elicit allergic reactions in susceptible individuals. Proper handling involves use of personal protective equipment such as gloves, lab coats, and eye protection, as well as ensuring adequate ventilation in working areas. Regulatory guidelines govern its use in cosmetic formulations to ensure consumer safety.
In summary, 6-amino-m-cresol is a chemically important aromatic compound valued mainly as an intermediate in hair dye production. Its combined amino, hydroxyl, and methyl functionalities confer reactivity that enables its use in oxidative dye chemistry and organic synthesis. Well-established synthetic methods and analytical characterization techniques support its industrial application in cosmetics and related chemical fields.
References
2024. Investigation of thermal properties and structural characterization of novel boron-containing Schiff base polymers. BMC Chemistry, 18(1).
DOI: 10.1186/s13065-024-01264-6
2021. Sesquicentennial birth anniversary of carbazole, a multifaceted wonder molecule: a revisit to its synthesis, photophysical and biological studies. Journal of the Iranian Chemical Society, 18(10).
DOI: 10.1007/s13738-021-02444-0
2012. Synthesis and theoretic calculations of benzoxazoles and docking studies of their interactions with triosephosphate isomerase. Medicinal Chemistry Research, 21(10).
DOI: 10.1007/s00044-012-0264-y
|

























































 GHS07 Warning Details
GHS07 Warning Details