Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Ji'nan Sheng Qi Medicine Science & Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shengqipharm.com | |||
 | +86 (531) 8889-2685 | |||
 | +86 (531) 8889-2171 | |||
 | 2153390475@qq.com | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| P and M Invest Ltd. | Russia | |||
|---|---|---|---|---|
 | www.fluorine.ru | |||
 | +7 (495) 135-6494 | |||
 | +7 (495) 135-6509 | |||
 | sales@fluorine1.ru | |||
| Chemical manufacturer | ||||
| Classification | API >> Anesthetic agents >> General anesthetics |
|---|---|
| Name | Sevoflurane |
| Synonyms | 1,1,1,3,3,3-Hexafluoro-2-(fluoromethoxy)-propane; Fluoromethyl 1,1,1,3,3,3-hexafluoroisopropyl ether |
| Molecular Structure | 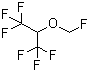 |
| Molecular Formula | C4H3F7O |
| Molecular Weight | 200.06 |
| CAS Registry Number | 28523-86-6 |
| EC Number | 643-089-7 |
| SMILES | C(OC(C(F)(F)F)C(F)(F)F)F |
| Density | 1.505 g/mL |
|---|---|
| Boiling point | 58 °C |
| Hazard Symbols |   GHS07;GHS08 Warning Details GHS07;GHS08 Warning Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H336-H361 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P318-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
Sevoflurane, a volatile liquid inhalation anesthetic, was discovered through systematic exploration in the field of pharmaceutical chemistry. It was first synthesized in the late 20th century as part of efforts to develop safer and more effective anesthetic agents. The discovery of Sevoflurane marked a significant milestone in anesthesia research, offering an alternative to existing volatile anesthetics with improved properties. Sevoflurane is widely used as an inhalation anesthetic during surgical procedures. Its rapid onset and offset of action, along with its minimal metabolism in the body, make it an ideal choice for inducing and maintaining anesthesia. Surgeons and anesthesiologists prefer Sevoflurane for its smooth induction and emergence, as well as its favorable hemodynamic stability during anesthesia. One of the notable applications of Sevoflurane is its use in pediatric anesthesia. Its pleasant odor, low airway irritation, and quick recovery profile make it particularly suitable for children undergoing surgery. Sevoflurane is often administered via inhalation through a mask or anesthesia machine, providing a smooth and comfortable anesthetic experience for pediatric patients. Sevoflurane's favorable pharmacokinetic profile and ease of administration make it well-suited for ambulatory or outpatient surgery settings. Patients undergoing minor procedures or surgeries that require short anesthesia durations benefit from Sevoflurane's rapid induction, rapid recovery, and minimal postoperative side effects. Its versatility and safety profile contribute to its widespread use in ambulatory surgical centers. In veterinary medicine, Sevoflurane is commonly employed as an inhalation anesthetic for various surgical procedures in animals. Its safety, efficacy, and ease of use make it a preferred choice among veterinarians for inducing and maintaining anesthesia in companion animals and livestock. Sevoflurane's quick recovery profile also allows for smooth transitions between anesthesia and consciousness in animals. Sevoflurane is increasingly being used in critical care settings, such as intensive care units (ICUs), for sedation and anesthesia management in mechanically ventilated patients. Its titratable effects and rapid elimination from the body make it a valuable tool for managing sedation levels and optimizing patient comfort and outcomes in the ICU setting. References 1998. Biological monitoring during exposure to the anesthetics isoflurane and sevoflurane. Zentralblatt fur Hygiene und Umweltmedizin. DOI: Not Available 1998. Postoperative nausea and vomiting. Der Anaesthesist. DOI: 10.1007/s001010050540 1998. High concentration versus incremental induction of anesthesia with sevoflurane in children: A comparison of induction times, vital signs, and complications. Journal of Clinical Anesthesia. DOI: 10.1016/s0952-8180(97)00218-3 |
| Market Analysis Reports |