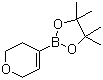
3,6-Dihydro-2H-pyran-4-boronic acid pinacol ester is an organic compound featuring a boronic acid ester functional group attached to a dihydropyran ring. Structurally, it consists of a six-membered oxygen-containing heterocyclic ring, known as dihydropyran, with partial saturation at positions 3 and 6, and a boronic acid pinacol ester substituent bonded at the 4-position of the ring. The pinacol ester is formed by the reaction of the boronic acid group with pinacol (2,3-dimethyl-2,3-butanediol), resulting in a cyclic boronate ester that enhances the compound’s stability and lipophilicity.
The dihydropyran ring serves as a common structural motif in organic chemistry, often employed in synthetic intermediates and natural product synthesis. Incorporation of the boronic acid pinacol ester functionality renders the molecule particularly useful in cross-coupling reactions, such as the Suzuki–Miyaura reaction, due to the well-known reactivity of boronates with organohalides catalyzed by palladium complexes.
Synthesis of 3,6-dihydro-2H-pyran-4-boronic acid pinacol ester typically involves the preparation of the corresponding boronic acid derivative, followed by esterification with pinacol under dehydrating conditions. Common synthetic routes include lithiation or metalation of halogenated dihydropyran precursors, subsequent borylation using boron reagents like bis(pinacolato)diboron, and ester formation through pinacol coordination.
The boronic acid pinacol ester moiety offers enhanced chemical stability compared to free boronic acids by protecting the boron center from oxidation and hydrolysis. This increased stability makes the compound convenient for handling, storage, and use in organic synthesis. Moreover, the ester group’s lipophilic nature facilitates solubility in organic solvents, which is advantageous in catalytic coupling reactions.
Applications of 3,6-dihydro-2H-pyran-4-boronic acid pinacol ester are primarily centered on its role as a versatile building block in organic synthesis. It is extensively used in palladium-catalyzed cross-coupling methodologies, particularly the Suzuki–Miyaura reaction, which forms carbon–carbon bonds between boronic esters and aryl or vinyl halides. Such reactions are widely employed in the synthesis of pharmaceuticals, agrochemicals, and complex natural products.
The compound’s heterocyclic ring and boronate ester functionalities allow the introduction of oxygen-containing heterocycles into molecular frameworks, which can modulate the physical and biological properties of target molecules. This makes it a valuable reagent for the construction of bioactive compounds and materials science applications.
Analytical characterization includes nuclear magnetic resonance (²H and ³C NMR) spectroscopy, which confirms the presence of the dihydropyran ring protons and carbons, as well as the pinacol methyl and methine groups. Boron NMR (B NMR) provides information about the boron environment, confirming ester formation. Infrared (IR) spectroscopy identifies characteristic absorptions related to the boronate ester and heterocyclic ether ring. Mass spectrometry verifies the molecular weight and fragmentation pattern consistent with the compound’s structure.
Physically, 3,6-dihydro-2H-pyran-4-boronic acid pinacol ester is generally a crystalline solid or viscous liquid depending on purity and preparation method. It exhibits good solubility in common organic solvents such as tetrahydrofuran (THF), dichloromethane, and ethyl acetate, but limited solubility in water due to its lipophilic ester groups.
In summary, 3,6-dihydro-2H-pyran-4-boronic acid pinacol ester is a boronate ester-functionalized oxygen heterocycle used extensively as a stable and reactive intermediate in palladium-catalyzed cross-coupling reactions. Its structure combines a dihydropyran ring with a pinacol-protected boronic acid, enabling efficient incorporation of heterocyclic motifs into complex organic molecules.
References
2019. Facile synthesis of novel amino acid-like building blocks by N-alkylation of heterocyclic carboxylates with N-Boc-3-iodoazetidine. Molecular Diversity, 24(4).
DOI: 10.1007/s11030-019-09987-8
2012. Synthesis by Miscellaneous Methods. Science of Synthesis.
URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-106-00047
|








































































 GHS07 Warning Details
GHS07 Warning Details