Online Database of Chemicals from Around the World
| Epochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.epochem.com | |||
 | +86 (21) 6760-1595 6760-1597 | |||
 | +86 (21) 6760-1605 | |||
 | seth_wang@epochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Jiangxi Time Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.groupchem.com | |||
 | +86 (794) 718-3888 718-3887 526-9970 +86 13867635890 | |||
 | +86 (794) 526-9668 / 526-9166 | |||
 | sales@groupchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Xinlian Rui Biomedical Technology (shandong) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xlrchem.com | |||
 | +86 15153173989 | |||
 | 3865799121@qq.com | |||
 | QQ Chat | |||
 | WeChat: 15153173989 | |||
| Chemical manufacturer since 2024 | ||||
| chemBlink Standard supplier since 2025 | ||||
| P and M Invest Ltd. | Russia | |||
|---|---|---|---|---|
 | www.fluorine.ru | |||
 | +7 (495) 135-6494 | |||
 | +7 (495) 135-6509 | |||
 | sales@fluorine1.ru | |||
| Chemical manufacturer | ||||
| Advance Research Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.fluoridearc.com | |||
 | +1 (918) 266-6789 | |||
 | +1 (918) 266-6796 | |||
 | sales@fluoridearc.com | |||
| Chemical manufacturer | ||||
| Gelest, Inc. | USA | |||
|---|---|---|---|---|
 | www.gelest.com | |||
 | +1 (215) 547-1015 | |||
 | +1 (215) 547-2484 | |||
 | info@gelest.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Organometallic compound >> Organic potassium |
|---|---|
| Name | Potassium trifluoroacetate |
| Synonyms | Trifluoroacetic acid potassium salt |
| Molecular Structure | 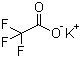 |
| Molecular Formula | C2F3KO2 |
| Molecular Weight | 152.11 |
| CAS Registry Number | 2923-16-2 |
| EC Number | 220-877-5 |
| SMILES | C(=O)(C(F)(F)F)[O-].[K+] |
| Density | 1.49 g/mL |
|---|---|
| Melting point | 140-142 °C |
| Hazard Symbols |   GHS06;GHS09 Danger Details GHS06;GHS09 Danger Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H300-H400-H412 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P264-P270-P273-P301+P316-P321-P330-P391-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
Potassium trifluoroacetate is an organofluorine salt composed of potassium cations and trifluoroacetate anions. It was first prepared in the mid-20th century as part of systematic investigations into fluorinated carboxylates, which were of growing interest because of their distinctive chemical and physical properties compared with non-fluorinated analogues. The trifluoromethyl group imparts strong electron-withdrawing character to the carboxylate, significantly modifying acidity, solubility, and reactivity. The salt form with potassium was developed to provide a stable, easy-to-handle source of the trifluoroacetate ion for use in chemical synthesis. The compound has been employed extensively as a reagent in organic chemistry. One of its primary applications is as a source of trifluoroacetate anions for nucleophilic substitution and elimination reactions. Because the trifluoromethyl group strongly stabilizes the negative charge of the carboxylate, potassium trifluoroacetate is less nucleophilic than other carboxylates but acts as an efficient leaving group and base under controlled conditions. This makes it valuable in reactions where a mild base is required. It has also been used in the synthesis of trifluoromethylated compounds, where the trifluoroacetate anion can serve as a precursor or auxiliary in the introduction of the trifluoromethyl group. In the field of polymer chemistry, potassium trifluoroacetate has been investigated as a reagent and catalyst component in fluoropolymer production. The incorporation of trifluoromethyl substituents in polymers improves thermal stability, chemical resistance, and hydrophobicity, properties that are critical in high-performance materials such as coatings, membranes, and specialty plastics. The salt’s solubility in polar solvents makes it suitable for homogeneous reaction systems, where it can act as a fluorinated additive or stabilizer. Another important application has been in analytical chemistry. Potassium trifluoroacetate is sometimes used in mass spectrometry as a volatile salt that facilitates ionization. Its decomposition yields volatile products, minimizing background signals and leaving little residue. This property has been exploited in peptide and protein analysis, where trifluoroacetate salts can improve sample preparation and ionization efficiency. In medicinal and agrochemical research, the compound has served as a building block for the synthesis of bioactive molecules. The trifluoromethyl group is widely used in drug design to alter pharmacokinetic properties such as lipophilicity and metabolic stability. Potassium trifluoroacetate, as a stable and convenient source of the trifluoroacetate ion, plays an indirect role in the creation of pharmaceutical and agrochemical agents where fluorinated functional groups are essential for biological performance. The discovery and continued use of potassium trifluoroacetate reflect the central importance of fluorinated reagents in modern chemistry. By providing a reliable means of introducing the trifluoroacetate moiety into synthetic schemes, it has contributed to advances across organic synthesis, materials science, and analytical chemistry. Its stability, solubility, and reactivity have made it a practical tool in the laboratory, ensuring its place among the widely utilized fluorine-containing salts in both research and applied chemistry. References 2022. Trifluoromethylation of Prefunctionalized Arenes or Alkenes. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-238-00138 2017. Photoelectrocatalytical Kolbe synthesis on thin film electrode of n-TiO2. Russian Journal of Electrochemistry, 53(2). DOI: 10.1134/s1023193517020070 2017. Hydration and dielectrical properties of aqueous pyrrolidinium trifluoroacetate solutions. Russian Journal of Physical Chemistry A, 91(10). DOI: 10.1134/s0036024417100247 |
| Market Analysis Reports |