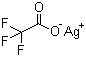
Silver trifluoroacetate (AgO2CCF3) is an organometallic compound consisting of a silver(I) cation and a trifluoroacetate anion. It is a white to off-white solid that is sensitive to light and moisture. The presence of the trifluoroacetate group imparts distinct solubility and reactivity characteristics compared to other silver salts, making it useful in various chemical transformations.
Silver trifluoroacetate has been employed as a reagent in organic synthesis, particularly in reactions involving halogen abstraction, oxidative coupling, and fluorination. It is known to serve as a mild oxidizing agent and a silver source in halide exchange reactions. One of its established applications is in the synthesis of fluoroorganic compounds, where it facilitates selective fluorination or trifluoromethylation by interacting with organohalides or other reactive substrates.
This compound has also been utilized in the preparation of silver-based catalysts and materials. Due to the high reactivity of silver ions, silver trifluoroacetate can be used as a precursor for silver nanoparticles, thin films, and coatings. These materials have applications in electronics, antimicrobial surfaces, and optical devices. Additionally, silver trifluoroacetate has been explored in coordination chemistry for the synthesis of metal-organic frameworks (MOFs) and other silver-containing complexes.
Handling and storage of silver trifluoroacetate require precautions due to its sensitivity to moisture and potential reactivity with organic compounds. It should be stored in a dry, light-protected environment to prevent decomposition. Like other silver salts, it may pose risks of skin and eye irritation, necessitating the use of appropriate protective equipment when handling.
The utility of silver trifluoroacetate in synthetic and material chemistry continues to be supported by its unique properties and established reactivity. Its role in fluorinated compound synthesis and silver-based material development highlights its importance in both academic and industrial research.
References
2021. Wonderful fusion of organofluorine chemistry and decarboxylation strategy. Chem. Soc. Rev., 50(10).
DOI: 10.1039/D1CS00216C
1952. The reactions of metallic salts of acids with halogens. Part II. The interaction of silver trifluoroacetate or silver perchlorate and halogens in various solvents. J. Chem. Soc..
DOI: 10.1039/JR9520000993
1951. The reactions of metallic salts of acids with halogens. Part I. The reaction of metal trifluoroacetates with iodine, bromine, and chlorine. J. Chem. Soc..
DOI: 10.1039/JR9510000584
|












































 GHS06;GHS07;GHS09 Danger Details
GHS06;GHS07;GHS09 Danger Details