Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Discovery Fine Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.discofinechem.com | |||
 | +44 (1202) 874-517 | |||
 | +44 (845) 094-4385 | |||
 | pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Extrasynthese Chemical S.A.S. | France | |||
|---|---|---|---|---|
 | www.extrasynthese.com | |||
 | +33 (47) 898-2034 | |||
 | +33 (47) 898-1945 | |||
 | info@extrasynthese.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Aktin Chemicals Inc | China | |||
|---|---|---|---|---|
 | www.aktinchem.com | |||
 | +86 400-028-7725 | |||
 | +86 (28) 8515-2372 | |||
 | info@aktinchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Chengdu Biopurify Phytochemicals Ltd. | China | |||
|---|---|---|---|---|
 | www.phytopurify.com | |||
 | +86 (28) 8263-3860 8263-3987 | |||
 | +86 (28) 8263-3165 | |||
 | sales@biopurify.com biopurify@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2017 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Cfm Oskar Tropitzsch GmbH | Germany | |||
|---|---|---|---|---|
 | www.cfmot.de | |||
 | +49 (9231) 9619-0 | |||
 | +49 (9231) 9619-60 | |||
 | info@cfmot.de | |||
| Chemical distributor since 1985 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Cayman Chemical Company | USA | |||
|---|---|---|---|---|
 | www.caymanchem.com | |||
 | +1 (734) 971-3335 | |||
 | +1 (734) 971-3640 | |||
 | sales@caymanchem.com | |||
| Chemical manufacturer | ||||
| Toronto Research Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.trc-canada.com | |||
 | +1 (416) 665-9696 | |||
 | +1 (416) 665-4439 | |||
 | info@trc-canada.com | |||
| Chemical manufacturer since 1982 | ||||
| Atlantic Research Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.atlantic-chemicals.com | |||
 | +44 (8707) 746-454 | |||
 | +44 (8707) 746-455 | |||
 | info@atlantic-chemicals.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | API >> Antiparasitic drug >> Anthelmintic medications |
|---|---|
| Name | Arecoline hydrobromide |
| Synonyms | Methyl 1,2,5,6-tetrahydro-1-methyl-3-pyridinecarboxylate hydrobromide |
| Molecular Structure | 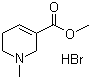 |
| Molecular Formula | C8H13NO2.HBr |
| Molecular Weight | 236.11 |
| CAS Registry Number | 300-08-3 |
| EC Number | 206-087-3 |
| SMILES | CN1CCC=C(C1)C(=O)OC.Br |
| Solubility | 47 mg/ mL (DMSO), 47 mg/ mL (water) |
|---|---|
| Melting point | 172 °C |
| Hazard Symbols |  GHS06 Danger Details GHS06 Danger Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301 Details | ||||||||||||||||||||
| Safety Statements | P264-P270-P301+P310+P330-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Arecoline hydrobromide, the hydrobromide salt of arecoline, is a notable chemical compound with diverse applications in pharmacology and medicine. Arecoline itself is a tropane alkaloid derived from the areca nut, commonly known as betel nut, which has been utilized for centuries in traditional medicine and cultural practices. The discovery of arecoline can be traced back to the study of the areca nut, which has a long history of use in various Asian cultures for its stimulant and psychoactive effects. Researchers identified arecoline as the principal active compound responsible for these effects. The hydrobromide salt form was developed to enhance the stability and solubility of arecoline, making it more suitable for pharmaceutical applications. The synthesis of arecoline hydrobromide involves the preparation of arecoline, which is typically extracted from the areca nut. The extraction process includes the isolation of arecoline through solvent extraction and purification methods. To form arecoline hydrobromide, arecoline is then reacted with hydrobromic acid. This reaction yields the hydrobromide salt, which improves the compound's solubility in aqueous solutions and facilitates its use in various formulations. In terms of application, arecoline hydrobromide has been studied for its potential therapeutic effects. Arecoline itself acts as a parasympathomimetic agent, meaning it mimics the effects of the parasympathetic nervous system. This activity has led to its investigation in treating conditions such as cognitive disorders, where its cholinergic properties may offer therapeutic benefits. Research has explored its potential role in enhancing cognitive function and memory. In addition to its pharmacological applications, arecoline hydrobromide has been used in scientific research to study the mechanisms of cholinergic neurotransmission. Its role as a cholinergic agent makes it a valuable tool in understanding how acetylcholine and related neurotransmitters function within the nervous system. This research is crucial for developing new treatments for neurological diseases and disorders. Furthermore, arecoline hydrobromide has applications in veterinary medicine, where it is used to investigate its effects on animal models. Its pharmacokinetics and effects on the autonomic nervous system are studied to assess its safety and efficacy in different species. These studies help in evaluating its potential for use in veterinary medicine and understanding its broader implications. Overall, arecoline hydrobromide represents a significant advancement in the utilization of arecoline, enhancing its solubility and potential for therapeutic use. Its role in pharmacological research and potential applications in both human and veterinary medicine underscore its importance as a chemical substance with diverse scientific and medical relevance. References 2024. Arecoline Hydrobromide Promotes the Apoptosis of Male Germ Cells. Cytology and Genetics, 58(5). DOI: 10.3103/s0095452724050128 2023. Arecoline hydrobromide suppresses PI3K/AKT pathway in rheumatoid arthritis synovial fibroblasts and relieves collagen-induced arthritis in mice. International Immunopharmacology, 124. DOI: 10.1016/j.intimp.2023.110925 1976. Improvement of the synthesis of arecoline from nicotinic acid. Pharmaceutical Chemistry Journal, 10(11). DOI: 10.1007/bf00760390 |
| Market Analysis Reports |