Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Epochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.epochem.com | |||
 | +86 (21) 6760-1595 6760-1597 | |||
 | +86 (21) 6760-1605 | |||
 | seth_wang@epochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Beijing Reco Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.recotechnology.com | |||
 | +86 (10) 6060-5164 | |||
 | +86 (10) 6060-5164 | |||
 | sales@recotechnology.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Labseeker Inc | USA | |||
|---|---|---|---|---|
 | www.labseeker.com | |||
 | +1 (858) 750-1632 | |||
 | +1 (858) 412-1220 | |||
 | marketing@labseeker.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Molcore Biopharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.molcore.com | |||
 | +86 (571) 8102-5280 | |||
 | sales@molcore.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Organo Fine Chemicals | India | |||
|---|---|---|---|---|
 | www.organo.in | |||
 | +91 (22) 2201-6968 2206-7521 | |||
 | +91 (22) 2205-9299 / 2208-3184 | |||
 | info@organo.in | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Hydrazine |
|---|---|
| Name | Methylhydrazine sulfate |
| Synonyms | Sulfuric acid |
| Molecular Structure | 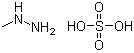 |
| Molecular Formula | CH6N2.H2SO4 |
| Molecular Weight | 144.14 |
| CAS Registry Number | 302-15-8 |
| EC Number | 206-115-4 |
| SMILES | CNN.OS(=O)(=O)O |
| Melting point | 143 °C (Expl.) |
|---|---|
| Hazard Symbols |  GHS06 Danger Details GHS06 Danger Details | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H300+H330-H311-H315-H319-H335-H351 Details | ||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P264-P280-P284 Details | ||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||
|
Methylhydrazine sulfate, with the chemical formula CH3NHNH2·H2SO4, is a chemical compound that has been studied for its properties and potential applications in various fields, particularly in the domains of rocket propulsion and pharmaceutical chemistry. It is a salt formed by the combination of methylhydrazine, a simple hydrazine derivative, and sulfuric acid. This compound is notable for its use in several industrial and chemical processes due to its reactive nature. Methylhydrazine itself was first synthesized in the early 20th century, with subsequent studies revealing its reactivity and utility in various chemical reactions. Its sulfate salt form, methylhydrazine sulfate, came to prominence for use in specialty chemical synthesis and as a propellant in aerospace applications. The compound is typically produced by neutralizing methylhydrazine with sulfuric acid, resulting in a stable crystalline salt. The primary application of methylhydrazine sulfate has historically been in the field of rocket propulsion. As a component in hypergolic propellant systems, it has been utilized as a fuel in various space missions. Hypergolic fuels, which ignite spontaneously upon contact with oxidizers, have been essential in many space programs due to their reliability and ease of use. Methylhydrazine sulfate, when used in combination with an oxidizer like nitrogen tetroxide, produces a highly efficient and stable reaction for use in spacecraft thrusters. Its high energy output and ability to maintain stability in extreme conditions make it a preferred choice for certain rocket engines, particularly in the mid-20th century during the Space Race era. In addition to its use in aerospace applications, methylhydrazine sulfate has also found a role in pharmaceutical and chemical research. As a derivative of hydrazine, it has been explored for its potential in the synthesis of various bioactive compounds, particularly those that can interact with biological systems in ways that may influence disease processes. Research into methylhydrazine sulfate has been conducted to understand its potential as a starting material in the synthesis of medicinal compounds, including those targeting cancer cells or other diseases that require specific chemical interactions. One important area of research involves the modification of hydrazine derivatives, such as methylhydrazine sulfate, for use in the development of anticancer agents. Hydrazine derivatives have demonstrated potential as inhibitors of specific enzymes and pathways involved in cancer cell growth, making methylhydrazine sulfate and related compounds an area of active interest for researchers working in medicinal chemistry. Despite its applications, methylhydrazine sulfate poses safety and environmental challenges due to the toxicity of hydrazine-based compounds. These substances are highly reactive and can be hazardous to both human health and the environment if not handled properly. As a result, there have been significant efforts to find safer alternatives to methylhydrazine sulfate in propulsion systems and other applications. The use of methylhydrazine sulfate has declined in favor of less toxic and more sustainable alternatives, but its historical importance in space exploration and chemical research remains significant. In conclusion, methylhydrazine sulfate is a versatile chemical compound with a variety of applications, most notably in the field of aerospace propulsion. Its role as a hypergolic fuel has contributed to the success of numerous space missions, while its use in pharmaceutical research has highlighted its potential as a precursor to bioactive compounds. Despite the challenges associated with its toxicity, methylhydrazine sulfate continues to be an important chemical substance in the history of space exploration and medicinal chemistry. References 2002. 2-Polyfluoroalkylchromones. 11. Synthesis and structures of 5-hydroxy-3-(2-hydroxyaryl)-5-polyfluoroalkyl-δ2-pyrazolines and 3(5)-(2-hydroxyaryl)-5(3)-polyfluoroalkylpyrazoles. Russian Chemical Bulletin, 51(7). DOI: 10.1023/a:1020960815497 1988. Study of the reaction of fluoroalkyl β-ketoesters with methylhydrazine by1H and19F NMR spectroscopy. Bulletin of the Academy of Sciences of the USSR, Division of chemical science, 37(2). DOI: 10.1007/bf00957434 1981. Condensed imidazo-1,2,4-azines. 4. Synthesis of 1,4-Dihydroimidazo[1,5-c]-1,2,4-triazine derivatives. Chemistry of Heterocyclic Compounds, 17(6). DOI: 10.1007/bf00503492 |
| Market Analysis Reports |