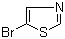
5-Bromothiazole is a five-membered heteroaromatic compound composed of a thiazole ring with a bromine atom substituted at the 5-position. Its molecular formula is C3H2BrNS, and it has a molecular weight of approximately 157.01 g/mol. The thiazole ring contains both a nitrogen atom at position 1 and a sulfur atom at position 3, creating an electron-rich heterocyclic system. The bromine atom at position 5 introduces a site for further functionalization while influencing the electronic distribution and steric profile of the ring.
The synthesis of 5-bromothiazole generally involves bromination of thiazole at the 5-position using controlled halogenation reactions. Direct bromination with bromine or N-bromosuccinimide under mild conditions allows selective substitution at the 5-position, guided by the electron density of the heterocycle. Alternative approaches may include the cyclization of α-bromo ketones with thiourea derivatives, which simultaneously constructs the thiazole ring and introduces the bromine atom in the desired position. Reaction conditions such as temperature, solvent, and stoichiometry are carefully optimized to minimize polybromination or undesired side reactions.
Chemically, 5-bromothiazole is moderately stable under ambient conditions and typically appears as a solid at room temperature. The bromine atom serves as an electrophilic site for metal-catalyzed cross-coupling reactions, including Suzuki, Sonogashira, or Buchwald–Hartwig couplings, enabling the introduction of a wide variety of substituents at the 5-position. The thiazole ring itself is aromatic and relatively electron-rich, making positions adjacent to the nitrogen and sulfur atoms moderately reactive toward electrophilic substitution under controlled conditions.
The compound exhibits solubility in polar and moderately nonpolar organic solvents such as dichloromethane, acetone, and ethanol. Its heteroaromatic structure allows participation in hydrogen bonding and π–π stacking interactions, which can influence crystallization, reactivity, and molecular recognition in both chemical and biological contexts. The combination of a reactive halogen and a stable heterocycle makes 5-bromothiazole an excellent intermediate for the synthesis of functionalized thiazole derivatives.
In practical applications, 5-bromothiazole is widely used in medicinal chemistry, agrochemical research, and organic synthesis as a versatile building block. Its bromine substituent allows for rapid diversification through cross-coupling reactions, while the thiazole core provides structural rigidity and electronic characteristics important for biological activity. The molecule is used to construct more complex heterocyclic scaffolds, bioactive molecules, and ligands, where selective functionalization at the 5-position is essential for tuning chemical and pharmacological properties.
Overall, 5-bromothiazole combines the chemical stability of the thiazole ring with the synthetic versatility of a halogen-substituted aromatic system. Its defined electronic and steric properties enable controlled transformations, making it a valuable intermediate for research and development in heterocyclic chemistry, drug discovery, and material science.
References
2025. Synthesis of 6-Amino-4-aryl-5-(4-arylthiazol-2-yl)-2-ethoxynicotinonitriles. Russian Journal of General Chemistry.
DOI: 10.1134/s1070363225601024
2023. Electrophilic MiniFrags Revealed Unprecedented Binding Sites for Covalent HDAC8 Inhibitors. Journal of Medicinal Chemistry.
DOI: 10.1021/acs.jmedchem.3c01779
2021. Synthesis and Structure of (2E)-3-Aryl(hetaryl)-2-[5-bromo-4-aryl(hetaryl)-1,3-thiazol-2-yl]acrylonitriles. Russian Journal of General Chemistry, 91(3).
DOI: 10.1134/s1070363221030038
|














































 GHS05;GHS07 Danger Details
GHS05;GHS07 Danger Details