Online Database of Chemicals from Around the World
| Polyunion Chemicals Limited | China | |||
|---|---|---|---|---|
 | www.polyunionchem.com | |||
 | +86 (133) 2649-6212 | |||
 | polyunionchem@outlook.com | |||
 | WhatsApp:+86-133-2649-6212 | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Chemical reagent >> Organic reagent >> Borate |
|---|---|
| Name | Tris(2-(2-(2-methoxyethoxy)ethoxy)ethyl) orthoborate |
| Synonyms | tris-(3,6,9-trioxadecyl) borate |
| Molecular Structure | 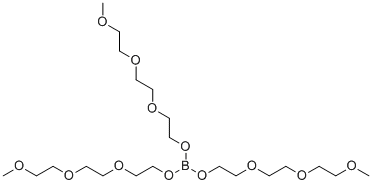 |
| Molecular Formula | C21H45BO12 |
| Molecular Weight | 500.39 |
| CAS Registry Number | 30989-05-0 |
| EC Number | 250-418-4 |
| SMILES | B(OCCOCCOCCOC)(OCCOCCOCCOC)OCCOCCOCCOC |
| Density | 1.1±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.436, Calc.* |
| Boiling Point | 493.3±45.0 °C (760 mmHg), Calc.* |
| Flash Point | 252.1±28.7 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS08 Warning Details GHS08 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H361fd Details | ||||||||||||||||
| Safety Statements | P203-P280-P318-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
|
Tris(2-(2-(2-methoxyethoxy)ethoxy)ethyl) orthoborate is a specialized organoboron compound featuring a boron atom coordinated to three ethoxyethyl groups, each containing a 2-(2-(2-methoxyethoxy)ethoxy) substituent. This unique structure endows the compound with distinctive chemical properties and has led to its use in various applications, particularly in the fields of materials science and catalysis. The discovery of tris(2-(2-(2-methoxyethoxy)ethoxy)ethyl) orthoborate stems from the need for boron-containing compounds with tailored reactivity and solubility characteristics. The compound’s structure is designed to enhance its stability and functionality by incorporating multiple ethoxy and methoxy groups, which improve its solubility in organic solvents and its compatibility with various reaction conditions. One of the primary applications of tris(2-(2-(2-methoxyethoxy)ethoxy)ethyl) orthoborate is in organic synthesis as a boron-based reagent. The compound is used as a Lewis acid catalyst in various reactions, including cross-coupling reactions and polymerizations. Its ability to stabilize intermediates and facilitate the formation of carbon-carbon bonds makes it a valuable tool in the synthesis of complex organic molecules. For example, it can be employed in the Suzuki-Miyaura coupling reaction, where it helps to form biaryl compounds by coupling arylboronic acids with aryl halides. The enhanced solubility and stability of the compound improve the efficiency and selectivity of these catalytic processes. In materials science, tris(2-(2-(2-methoxyethoxy)ethoxy)ethyl) orthoborate is utilized in the development of advanced materials, particularly those involving polymer chemistry. The compound’s boron-oxygen framework can be incorporated into polymer matrices to enhance their mechanical and thermal properties. For instance, it can be used as a cross-linking agent in the synthesis of high-performance polymers or as a component in the preparation of boron-doped materials with specific electronic or optical properties. The incorporation of the compound into polymer systems can lead to materials with improved durability, thermal stability, and chemical resistance. Another significant application of tris(2-(2-(2-methoxyethoxy)ethoxy)ethyl) orthoborate is in the field of fluorescent sensors and imaging. The compound can be designed to form fluorescent complexes with specific analytes, making it useful for detecting and quantifying various substances. Its ability to form stable complexes with certain ions or molecules allows for the development of sensitive and selective sensors for environmental monitoring, medical diagnostics, and analytical chemistry. The incorporation of the compound into sensor materials enhances their performance by providing high sensitivity and selectivity. Overall, the unique chemical structure of tris(2-(2-(2-methoxyethoxy)ethoxy)ethyl) orthoborate provides several advantages in its applications. Its stability, solubility, and reactivity make it a versatile reagent in organic synthesis and materials science, while its potential for use in fluorescent sensing expands its utility in various analytical applications. Continued research into this compound and its derivatives will likely lead to further innovations and applications in chemistry and materials science. References 1995. Fractional analysis of technical polyoxaalkyl borates by TG. Journal of Thermal Analysis and Calorimetry, 44(1). DOI: 10.1007/bf02635980 2006. Polyethylene glycol monomethyl ethers as the main component of brake fluid. Russian Journal of Applied Chemistry, 79(11). DOI: 10.1134/s107042720611022x 2009. Synthesis and Modeling of Polysiloxane-Based Salt-in-Polymer Electrolytes with Various Additives. The Journal of Physical Chemistry B, 113(43). DOI: 10.1021/jp907832q |
| Market Analysis Reports |