Online Database of Chemicals from Around the World
| Zhejiang Yangfan New Materials Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shoufu.com | |||
 | +86 (571) 8890-2510 8890-2504 8890-2511 | |||
 | +86 (571) 8890-2500 | |||
 | shoufu@shoufuchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Molekula Ltd | UK | |||
|---|---|---|---|---|
 | www.molekula.com | |||
 | +44 (1747) 831-066 | |||
 | +44 (1747) 831-199 | |||
 | info@molekula.com | |||
| Chemical manufacturer | ||||
| Taizhou Skyway Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.skywaychem.com | |||
 | +86 (576) 8547-1369 | |||
 | +86 (576) 8257-2096 | |||
 | skyway@skywaychem.com | |||
| Chemical manufacturer | ||||
| Toronto Research Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.trc-canada.com | |||
 | +1 (416) 665-9696 | |||
 | +1 (416) 665-4439 | |||
 | info@trc-canada.com | |||
| Chemical manufacturer since 1982 | ||||
| Oakwood Products, Inc. | USA | |||
|---|---|---|---|---|
 | www.oakwoodchemical.com | |||
 | +1 (800) 467-3386 | |||
 | k_weaver@oakwoodchemical.com | |||
| Chemical manufacturer | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Thiophene compound |
|---|---|
| Name | 2,5-Dibromothiophene |
| Molecular Structure | 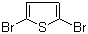 |
| Molecular Formula | C4H2Br2S |
| Molecular Weight | 241.93 |
| CAS Registry Number | 3141-27-3 |
| EC Number | 221-547-3 |
| SMILES | C1=C(SC(=C1)Br)Br |
| Water solubility | practically insoluble |
|---|---|
| Density | 2.2±0.1 g/cm3, Calc.*, 2.147 g/mL (Expl.) |
| Melting point | -6 °C (Expl.) |
| Index of Refraction | 1.638, Calc.*, 1.629 (Expl.) |
| Boiling Point | 209.5±20.0 °C (760 mmHg), Calc.*, 211 °C (Expl.) |
| Flash Point | 99.4±0.0 °C, Calc.*, 99 °C (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS06;GHS07;GHS08 Danger Details GHS06;GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H300-H315-H317-H319-H335-H341 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P261-P264-P264+P265-P270-P271-P272-P280-P301+P316-P302+P352-P304+P340-P305+P351+P338-P318-P319-P321-P330-P332+P317-P333+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||
|
2,5-Dibromothiophene is an organic compound that consists of a thiophene ring, a sulfur-containing five-membered heterocycle, with bromine atoms attached to the 2 and 5 positions. Its chemical formula is C4H2Br2S, and it is classified as a halogenated thiophene derivative. The compound appears as a yellow to light brown crystalline solid and is primarily used in organic synthesis, particularly in the preparation of materials with semiconducting properties. The discovery of 2,5-dibromothiophene can be traced to the development of halogenated thiophenes as versatile building blocks in organic chemistry. Thiophene, first discovered in the 19th century, has since become a key heterocyclic compound due to its aromaticity and ability to participate in various reactions, such as electrophilic substitution. The addition of halogens like bromine to the thiophene ring enhances its reactivity and opens up new pathways for functionalizing organic materials, making it valuable in diverse applications. 2,5-Dibromothiophene has several significant applications, particularly in the field of organic electronics. It is commonly used in the synthesis of organic semiconductors, which are materials that can conduct electricity and are employed in the production of organic light-emitting diodes (OLEDs), organic photovoltaic cells (OPVs), and organic field-effect transistors (OFETs). The bromine atoms in 2,5-dibromothiophene play an essential role in enhancing the solubility and stability of the resulting materials, which is crucial for their performance in electronic devices. One of the most notable applications of 2,5-dibromothiophene is in the preparation of conjugated polymers. These polymers are used in a variety of electronic and optoelectronic devices, where their conductivity and optical properties are tailored by altering the monomers from which they are derived. 2,5-Dibromothiophene serves as a key monomer for the synthesis of poly(thiophene) derivatives, which have been widely used in the development of flexible electronics, solar cells, and sensors. In addition to electronics, 2,5-dibromothiophene is also utilized in the synthesis of other functionalized organic compounds. For example, it is used in the preparation of certain dyes, pigments, and intermediates in the production of pharmaceuticals. Its reactivity makes it an important precursor in the design of new materials with specialized properties for applications in various chemical and industrial sectors. The compound’s role as a building block in organic synthesis, particularly for materials used in electronics, continues to drive research into its applications. Ongoing studies focus on improving the efficiency and stability of devices made from 2,5-dibromothiophene derivatives, with the aim of enhancing the performance of organic electronics in commercial applications. References 2020. Trapping of Transient Thienyllithiums Generated by Deprotonation of 2,3- or 2,5-Dibromothiophene in a Flow Microreactor. Synlett, 31(18). DOI: 10.1055/s-0040-1706479 2018. Classes of CPs: Part 2. Conducting Polymers, Fundamentals and Applications. DOI: 10.1007/978-3-319-69378-1_35 2016. A novel double-layer electrospun nanofibrous membrane sensor for detecting nitroaromatic compounds. Journal of Materials Science, 51(19). DOI: 10.1007/s10853-016-0252-6 |
| Market Analysis Reports |