Online Database of Chemicals from Around the World
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Classification | Chemical reagent >> Organic reagent >> Phosphine ligand |
|---|---|
| Name | Bis-(3-nitrophenyl)-methylphosphine oxide |
| Synonyms | 1-[methyl-(3-nitrophenyl)phosphoryl]-3-nitrobenzene |
| Molecular Structure | 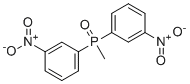 |
| Molecular Formula | C13H11N2O5P |
| Molecular Weight | 306.21 |
| CAS Registry Number | 31638-90-1 |
| EC Number | 653-964-5 |
| SMILES | CP(=O)(C1=CC=CC(=C1)[N+](=O)[O-])C2=CC=CC(=C2)[N+](=O)[O-] |
| Density | 1.4±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.611, Calc.* |
| Boiling Point | 445.6±30.0 °C (760 mmHg), Calc.* |
| Flash Point | 223.3±24.6 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302 Details | ||||||||
| Safety Statements | P264-P270-P301+P317-P330-P501 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
|
Bis-(3-nitrophenyl)-methylphosphine oxide is a notable chemical compound with significant applications in organophosphorus chemistry and catalysis. This substance features a phosphorus atom bonded to two 3-nitrophenyl groups and a methyl group, with an oxide group attached to the phosphorus. Its structure endows it with distinctive chemical properties that make it valuable in various synthetic applications. The discovery of Bis-(3-nitrophenyl)-methylphosphine oxide was motivated by the need for versatile phosphine oxide ligands in organometallic chemistry. The 3-nitrophenyl groups provide electronic and steric effects that influence the ligand's interaction with metal centers. The presence of the nitro group enhances the electron-withdrawing characteristics, which can stabilize certain metal complexes and modulate their reactivity. One of the primary applications of Bis-(3-nitrophenyl)-methylphosphine oxide is in catalysis, particularly in metal-catalyzed reactions. The ligand is employed to stabilize metal complexes in various catalytic transformations, including cross-coupling reactions, hydrogenation, and oxidation processes. In these reactions, the ligand's electronic properties play a crucial role in influencing the reactivity and selectivity of the metal catalyst. The compound has also found applications in the development of novel catalysts for organic synthesis. Its ability to form stable complexes with transition metals allows for the design of new catalytic systems that can perform specific chemical transformations with high efficiency. The use of such ligands has expanded the scope of organometallic catalysis, enabling the synthesis of complex molecules and materials. Moreover, Bis-(3-nitrophenyl)-methylphosphine oxide is valuable in the study of reaction mechanisms and catalyst design. By altering the electronic environment around the metal center, researchers can gain insights into the factors that control catalytic activity and selectivity. This information is crucial for optimizing catalytic processes and developing more efficient synthetic methods. In summary, Bis-(3-nitrophenyl)-methylphosphine oxide is an important ligand in organometallic chemistry, known for its role in stabilizing metal complexes and enhancing catalytic processes. Its unique structure and properties make it a valuable tool for researchers and chemists involved in the development of new catalysts and synthetic methodologies. References none |
| Market Analysis Reports |