Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Shanghai Daji Medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.djpharm.com | |||
 | +86 13816245196 | |||
 | sales1@djpharm.com | |||
| Chemical distributor since 2019 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Atlantic Research Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.atlantic-chemicals.com | |||
 | +44 (8707) 746-454 | |||
 | +44 (8707) 746-455 | |||
 | info@atlantic-chemicals.com | |||
| Chemical manufacturer | ||||
| Activate Scientific GmbH | Germany | |||
|---|---|---|---|---|
 | www.activate-scientific.com | |||
 | +49 (8051) 965-1660 | |||
 | +49 (8051) 965-1661 | |||
 | rschmitt@activate-scientific.com | |||
| Chemical manufacturer | ||||
| Spectrum Info Ltd. | Ukraine | |||
|---|---|---|---|---|
 | www.spec-info.com | |||
 | +38 (044) 573-5462 | |||
 | +38 (044) 573-2397 | |||
 | spec-info@spec-info.com | |||
| Chemical manufacturer | ||||
| Matrix Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.matrixscientific.com | |||
 | +1 (803) 788-9494 | |||
 | +1 (803) 788-9419 | |||
 | sales@matrixscientific.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Amino acids and their derivatives >> Ester amino acid |
|---|---|
| Name | Ethyl 2-aminothiophene-3-carboxylate |
| Synonyms | 2-Aminothiophene-3-carboxylic acid ethyl ester |
| Molecular Structure | 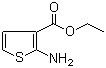 |
| Molecular Formula | C7H9NO2S |
| Molecular Weight | 171.22 |
| CAS Registry Number | 31891-06-2 |
| EC Number | 672-859-5 |
| SMILES | CCOC(=O)C1=C(SC=C1)N |
| Density | 1.3±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 42 - 44 °C (Expl.) |
| Boiling point | 273.6±20.0 °C 760 mmHg (Calc.)* |
| Flash point | 119.3±21.8 °C (Calc.)* |
| Index of refraction | 1.582 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Ethyl 2-aminothiophene-3-carboxylate is a heteroaromatic compound in which a thiophene ring is substituted with an amino group at position 2 and an ethyl ester at position 3. Its molecular formula is C7H9NO2S, with a molecular weight of approximately 167.22 g/mol. The thiophene ring is a five-membered sulfur-containing aromatic system, which provides electron-rich characteristics, while the amino and ester substituents impart both nucleophilicity and electrophilicity, enabling diverse chemical transformations. The compound is commonly synthesized via cyclization strategies that construct the thiophene core with pre-installed substituents. One approach involves the reaction of α-haloketones or 1,3-dicarbonyl compounds with sulfur sources and amines to generate the 2-aminothiophene skeleton. Subsequent esterification at position 3 produces the ethyl ester derivative. Alternative methods include condensation of aminothiophene intermediates with ethyl chloroformate or other activated carboxylating agents to selectively install the ester functionality. Reaction conditions are optimized to preserve the amino group and avoid overreaction or decomposition of the thiophene ring. Chemically, ethyl 2-aminothiophene-3-carboxylate displays reactivity at both the amino and ester positions. The amino group can participate in acylation, alkylation, or condensation reactions, while the ester is amenable to hydrolysis, transesterification, or amidation. The electron-rich thiophene ring can undergo electrophilic substitution reactions, particularly at positions 4 and 5, although the presence of the substituents at positions 2 and 3 influences regioselectivity. The combination of these functional groups allows for selective derivatization while maintaining the integrity of the heteroaromatic core. The compound is typically a solid at room temperature with moderate solubility in polar organic solvents such as ethanol, acetone, and dimethylformamide. Its stability under ambient conditions is adequate, though it should be protected from strong oxidizing agents and acids or bases that could hydrolyze the ester or decompose the amino group. The molecule’s structure, combining a reactive amino group and a heteroaromatic ring, facilitates hydrogen bonding and π–π stacking, which can influence crystallization and intermolecular interactions. In practical applications, ethyl 2-aminothiophene-3-carboxylate serves as a versatile intermediate in medicinal chemistry and organic synthesis. It can be used to prepare a variety of thiophene-based derivatives, including amides, heterocycles, and bioactive molecules. The combination of an amino group and ester functionality provides multiple points for modification, enabling the exploration of structure–activity relationships in pharmaceuticals, agrochemicals, and materials science. Overall, the combination of a sulfur-containing heterocyclic ring, an amino substituent, and an ester group gives ethyl 2-aminothiophene-3-carboxylate its synthetic versatility. Its defined reactivity and functional handles make it a valuable building block for the construction of complex heteroaromatic compounds, supporting applications in chemical research, drug development, and advanced organic synthesis. References 2015. Ethyl 2-Aminothiophene-3-Carboxylates in the Synthesis of Isomeric Thienopyridines. Chemistry of Heterocyclic Compounds, 50(10). DOI: 10.1007/s10593-015-1647-z 2008. A New One-Pot Route for the Synthesis of Fused Pyrido[1,2-a]pyrimidin-4-ones. Synthesis. DOI: 10.1055/s-2008-1066988 2007. Ethyl 2-aminothiophene-3-carboxylate. Acta Crystallographica Section E, 63(4). DOI: 10.1107/s1600536807009683 |
| Market Analysis Reports |