Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Hebei Duling International Trad Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hbduling.cn | |||
 | +86 15227159736 | |||
 | sales04@hbduling.cn | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Toronto Research Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.trc-canada.com | |||
 | +1 (416) 665-9696 | |||
 | +1 (416) 665-4439 | |||
 | info@trc-canada.com | |||
| Chemical manufacturer since 1982 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Carboxylic acid |
|---|---|
| Name | 2-(4-Bromophenyl)-2-methylpropionic acid |
| Synonyms | 2-Methyl-2-(4-bromophenyl)propionic acid |
| Molecular Structure | 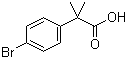 |
| Molecular Formula | C10H11BrO2 |
| Molecular Weight | 243.10 |
| CAS Registry Number | 32454-35-6 |
| EC Number | 696-555-7 |
| SMILES | CC(C)(C1=CC=C(C=C1)Br)C(=O)O |
| Melting point | 122-124 °C |
|---|---|
| Boiling Point | 334.239 °C (760 mmHg) |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302 Details | ||||||||||||||||||||||||
| Safety Statements | P264-P270-P301+P317-P330-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
2-(4-Bromophenyl)-2-methylpropionic acid, also known as 4-bromo-α-methyl-2-phenylpropionic acid, is an important compound in the field of organic chemistry. Its chemical structure comprises a bromine atom and a phenyl group attached to a propionic acid backbone. The compound's molecular formula is C11H13BrO2. The discovery of 2-(4-Bromophenyl)-2-methylpropionic acid is rooted in the broader research into halogenated aromatic compounds and their derivatives. Researchers have long recognized the utility of halogenated phenyl groups in organic synthesis due to their ability to participate in a range of chemical reactions. The introduction of a bromine atom into the phenyl ring, combined with the presence of a methyl group on the propionic acid moiety, endows the molecule with unique chemical properties that are valuable for various applications. In terms of its synthetic applications, 2-(4-Bromophenyl)-2-methylpropionic acid serves as a key intermediate in the synthesis of pharmaceuticals and agrochemicals. The bromine atom in the phenyl ring provides a site for further chemical modifications, allowing the introduction of additional functional groups or the formation of complex molecular structures. This makes the compound a valuable building block in the synthesis of biologically active molecules. One notable application of 2-(4-Bromophenyl)-2-methylpropionic acid is in the development of nonsteroidal anti-inflammatory drugs (NSAIDs). The compound's structure is related to several NSAIDs, including ibuprofen, a widely used medication for pain and inflammation. The bromine atom in 2-(4-Bromophenyl)-2-methylpropionic acid can be utilized to introduce variations in the NSAID structure, leading to the development of new compounds with potentially improved efficacy or reduced side effects. In addition to its pharmaceutical applications, 2-(4-Bromophenyl)-2-methylpropionic acid is also used in the field of materials science. The compound's ability to participate in chemical reactions makes it a useful precursor for the synthesis of functionalized polymers and materials. By modifying the compound's structure, researchers can create materials with specific properties for applications in areas such as electronics, coatings, and sensors. The compound's role in chemical research extends to its use as a reagent in various chemical transformations. For example, 2-(4-Bromophenyl)-2-methylpropionic acid can be employed in cross-coupling reactions, where it acts as an electrophile to form new carbon-carbon bonds. This capability is crucial for the construction of complex organic molecules and the development of new chemical methodologies. Overall, 2-(4-Bromophenyl)-2-methylpropionic acid represents a significant advancement in the field of organic chemistry. Its discovery and applications underscore its importance as a versatile building block in the synthesis of pharmaceuticals, materials, and other chemical compounds. The compound's unique properties and reactivity make it a valuable tool for researchers and chemists working in various areas of chemical science. References 2016. Alectinib. Pharmaceutical Substances, 2016. 2015. Intramolecular C-O Bond Formation. Science of Synthesis, 2015, 2, 365. DOI: 10.1055/sos-SD-218-00206 |
| Market Analysis Reports |