Online Database of Chemicals from Around the World
| Ereztech LLC | USA | |||
|---|---|---|---|---|
 | www.ereztech.com | |||
 | +1 (888) 658-1221 | |||
 | sales@ereztech.com | |||
| Chemical distributor since 2010 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Intatrade Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.intatrade.de | |||
 | +49 (3493) 605-465 | |||
 | +49 (3493) 605-470 | |||
 | sales@intatrade.de | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Fujian Wolfa Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.wolfabio.com | |||
 | +86 18050950397 | |||
 | jomin@wolfabio.com | |||
 | WhatsApp:+86 18359103607 | |||
| Chemical manufacturer since 2023 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Dalchem | Russia | |||
|---|---|---|---|---|
 | www.dalchem.com | |||
 | +7 (8312) 753-772 | |||
 | +7 (8312) 750-799 | |||
 | dregichy@dalchem.com | |||
| Chemical manufacturer since 1997 | ||||
| Gelest, Inc. | USA | |||
|---|---|---|---|---|
 | www.gelest.com | |||
 | +1 (215) 547-1015 | |||
 | +1 (215) 547-2484 | |||
 | info@gelest.com | |||
| Chemical manufacturer | ||||
| Boulder Scientific Company | USA | |||
|---|---|---|---|---|
 | www.bouldersci.com | |||
 | +1 (970) 535-4494 | |||
 | +1 (970) 535-4584 | |||
 | sales@bouldersci.com | |||
| Chemical manufacturer | ||||
| ProChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.prochemonline.com | |||
 | +1 (815) 398-1788 | |||
 | +1 (815) 398-1810 | |||
 | prochem3@aol.com | |||
| Chemical manufacturer since 1986 | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Organometallic compound >> Organic titanium |
|---|---|
| Name | Titanium dimethylamide |
| Synonyms | Titanium tetrakis(dimethylamide); Tetrakis(dimethylamino)titanium |
| Molecular Structure | 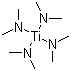 |
| Molecular Formula | C8H24N4Ti |
| Molecular Weight | 224.17 |
| CAS Registry Number | 3275-24-9 |
| EC Number | 221-904-3 |
| SMILES | C[N-]C.C[N-]C.C[N-]C.C[N-]C.[Ti+4] |
| Density | 0.947 |
|---|---|
| Boiling point | 50 °C (0.5 mmHg) |
| Flash point | -5 °C |
| Hazard Symbols |    GHS02;GHS05;GHS06'GHS07 Danger Details GHS02;GHS05;GHS06'GHS07 Danger Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H225-H260-H302+H332-H302-H314-H318-H331-H332 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P210-P223-P231+P232-P233-P240-P241-P242-P243-P260-P261-P264-P264+P265-P270-P271-P280-P301+P317-P301+P330+P331-P302+P335+P334-P302+P361+P354-P303+P361+P353-P304+P340-P305+P354+P338-P316-P317-P321-P330-P363-P370+P378-P402+P404-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| Transport Information | UN 3398 | ||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
Titanium dimethamide, with the chemical formula Ti(NMe2)2, is an organotitanium compound that plays an important role in materials science and chemical synthesis. This compound, which has a titanium atom coordinated by two dimethamide groups, has attracted much attention due to its unique chemical properties and wide range of applications. The discovery of titanium dimethamide is associated with the advancement of transition metal amide chemistry. The synthesis of Ti(NMe2)2 involves the reaction of titanium tetrachloride with dimethylamine to form this dimethamide complex. The structure of this compound generally features a titanium center and two dimethamide ligands arranged in a geometric shape that imparts specific reactivity and stability characteristics. One of the main applications of titanium dimethamide is in the field of chemical vapor deposition (CVD). The compound can be used as a precursor for the deposition of titanium-based thin films and coatings. These films are essential for a variety of applications in electronics and materials engineering, where properties such as titanium's high strength and corrosion resistance are advantageous. Ti(NMe2)2 is able to form high-quality titanium films, thereby improving the performance and durability of electronic devices, including semiconductors and sensors. In addition to its role in CVD, titanium dimethamide is used as a catalyst or catalyst precursor in chemical reactions. The compound can facilitate a variety of transformations, including polymerization and cross-coupling reactions. Its ability to stabilize reaction intermediates and facilitate specific chemical processes makes it a valuable tool in both industrial and research settings. The unique electronic and stereogenic properties of Ti(NMe2)2 allow for increased effectiveness as a catalyst in a variety of chemical applications. Titanium dimethamide has also been used to study transition metal amides and their interactions with other ligands. Researchers have used this compound to gain insight into how titanium behaves in different chemical environments and to develop new materials with tailored properties. The knowledge gained from studying Ti(NMe2)2 has enhanced the understanding of coordination chemistry and the design of advanced materials for a variety of applications. Due to the reactive nature of titanium dimethamide and the potential for association with dimethamide groups, standard safety precautions need to be taken when handling titanium dimethamide. Proper safety practices, including the use of personal protective equipment and working in well-ventilated areas, are essential to ensure safe handling and storage of this compound. Ongoing research continues to explore new applications and improve the use of titanium dimethamide in different fields. Advances in materials science, catalysis, and thin-film technology are expected to expand the use of this compound and lead to further innovations. In summary, titanium dicarbonamide is an important organotitanium compound with important applications in chemical vapor deposition, catalysis, and research. Its discovery and development have led to advances in materials science and chemical processes, and continued research is expected to reveal more uses and improvements. References 2020. Catalysts via Atomic Layer Deposition. Recent Advances in Nanoparticle Catalysis, Not applicable. DOI: 10.1007/978-3-030-45823-2_3 1990. Titanium Nitride Thin Films: Properties and APCVD Synthesis Using Organometallic Precursors. MRS Online Proceedings Library, 168. DOI: 10.1557/proc-168-357 2021. Tetrakis(dimethylamino)titanium(IV)-Mediated Dimerization of Azomethine Ylides. Science of Synthesis, Not applicable. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-140-00349 |
| Market Analysis Reports |