Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Manus Aktteva | India | |||
|---|---|---|---|---|
 | www.manusaktteva.in | |||
 | +91 (79) 6512-3395 | |||
 | +91 (79) 2646-3395 | |||
 | products@manusakttevabiopharma.in | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2008 | ||||
| Quzhou HuntBio Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.huntchem.com | |||
 | +86 (571) 6406-7827 +86 13868119375 | |||
 | +86 (571) 6479-8288 | |||
 | sales@huntchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2009 | ||||
| TH Chemica Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.thchemica.com | |||
 | +86 (573) 8283-5115 +86 13998887555 | |||
 | +86 (573) 8396-5652 | |||
 | thchemica@126.com info@thchemica.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Porphyrin Systems | Germany | |||
|---|---|---|---|---|
 | www.porphyrin-systems.de | |||
 | +49 (40) 6466-5860 | |||
 | +49 (40) 6466-5861 | |||
 | hombrecher@porphyrin-systems.de | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shandong Boyuan Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.boyuanpharm.com | |||
 | +86 (531) 6995-4981 8896-3280 +86 15806417970 | |||
 | +86 (531) 8896-4879 | |||
 | Jeffrey.Liu@boyuanpharm.com boyuanchem@126.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Qichuang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qc-chemical.com | |||
 | +86 (571) 8893-5129 | |||
 | +86 (571) 8825-0182 | |||
 | david@qc-chemical.com sales@qc-chemical.com davidw0828@gmail.com | |||
 | QQ Chat | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Eastar Chemical Corporation | USA | |||
|---|---|---|---|---|
 | www.eastarchem.com | |||
 | +1 800-898-2436 | |||
 | +1 (877) 898-2436 | |||
 | info@eastarchem.com | |||
| Chemical manufacturer since 1989 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Shanghai Edu Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemido.com | |||
 | +86 19301251526 | |||
 | sales1@chemido.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Georganics Ltd. | Slovakia | |||
|---|---|---|---|---|
 | www.georganics.sk | |||
 | +421 (33) 640-3132 | |||
 | +421 (33) 640-3133 | |||
 | georganics@georganics.sk | |||
| Chemical manufacturer since 1998 | ||||
| Ryan Scientific, Inc. | USA | |||
|---|---|---|---|---|
 | www.ryansci.com | |||
 | +1 (843)-884-4911 | |||
 | +1 (843) 884-5568 | |||
 | sales@ryansci.com | |||
| Chemical manufacturer | ||||
| Sinbond Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinbondpharm.com | |||
 | +86 (531) 8703-8285 +86 13583181986 +44 (208) 242-5518 | |||
 | +86 (531) 8703-8285 | |||
 | cici1124@gmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| Advanced Chemical Intermediates Ltd. | UK | |||
|---|---|---|---|---|
 | www.acints.com | |||
 | +44 (1840) 261-451 | |||
 | +44 (1840) 261-451 | |||
 | enquiries@acints.com | |||
| Chemical manufacturer | ||||
| Synthonix, Inc. | USA | |||
|---|---|---|---|---|
 | www.synthonix.com | |||
 | +1 (919) 875-9277 | |||
 | +1 (919) 875-9601 | |||
 | info@synthonix.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives >> Carboxylic esters and their derivatives |
|---|---|
| Name | Ethyl 2-aminothiazole-5-carboxylate |
| Synonyms | Ethyl 2-amino-1,3-thiazole-5-carboxylate |
| Molecular Structure | 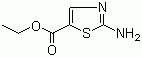 |
| Molecular Formula | C6H8N2O2S |
| Molecular Weight | 172.20 |
| CAS Registry Number | 32955-21-8 |
| EC Number | 676-105-6 |
| SMILES | CCOC(=O)C1=CN=C(S1)N |
| Density | 1.3±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 159 - 163 °C (Expl.) |
| Boiling point | 308.0±15.0 °C 760 mmHg (Calc.)* |
| Flash point | 140.1±20.4 °C (Calc.)* |
| Index of refraction | 1.588 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302+H312+H332-H302-H312-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
Ethyl 2-aminothiazole-5-carboxylate is a heteroaromatic compound consisting of a five-membered thiazole ring substituted with an amino group at position 2 and an ethyl ester at position 5. Its molecular formula is C6H8N2OS, with a molecular weight of approximately 144.20 g/mol. The thiazole ring contains both a nitrogen atom at position 1 and a sulfur atom at position 3, creating an electron-rich heterocyclic system. The amino and ester substituents confer both nucleophilic and electrophilic reactivity, making the molecule suitable for a range of chemical transformations. The synthesis of ethyl 2-aminothiazole-5-carboxylate typically involves the construction of the thiazole ring from α-haloketones or 1,3-dicarbonyl compounds with a sulfur source and a nitrogen-containing reagent. The amino group at position 2 is usually introduced during ring formation using ammonia or a primary amine, while the ester functionality at position 5 can be installed either through direct cyclization with an ester-containing precursor or via subsequent esterification of a carboxylic acid. Careful control of reaction conditions, including temperature, solvent, and stoichiometry, is required to achieve selective formation of the 2-amino and 5-ester substitution pattern. Chemically, the amino group is nucleophilic and can undergo acylation, alkylation, or condensation reactions, while the ethyl ester is reactive toward hydrolysis, transesterification, or amidation. The electron-rich thiazole ring allows for electrophilic substitution, particularly at positions 4 and 5, but the presence of substituents at positions 2 and 5 modifies the regioselectivity. This combination of functional groups provides multiple points of chemical modification while preserving the heteroaromatic core. The compound is generally a solid at room temperature and demonstrates solubility in polar organic solvents such as ethanol, acetone, and dimethylformamide. It is chemically stable under ambient conditions but should be protected from strong oxidizing agents, acids, or bases that could decompose the ester or amino functionalities. The molecule’s structure enables hydrogen bonding and π–π interactions, which can influence crystallization, solubility, and reactivity in synthetic applications. In practical applications, ethyl 2-aminothiazole-5-carboxylate is widely used as a building block in medicinal chemistry, agrochemical research, and heterocyclic synthesis. The amino and ester groups allow for selective functionalization, making it suitable for the preparation of thiazole-based derivatives, heterocyclic scaffolds, and bioactive compounds. It is particularly valuable in the synthesis of small molecules designed for biological activity, where the thiazole ring provides structural rigidity and electronic properties relevant to target binding. Overall, the combination of a thiazole ring, a nucleophilic amino group, and an electrophilic ester group renders ethyl 2-aminothiazole-5-carboxylate a versatile intermediate. Its predictable reactivity and multiple functional handles support its use in organic synthesis, drug discovery, and the development of complex heterocyclic compounds. References 2010. From Ethyl 3-Ethoxyacrylate and Thioureas. Science of Synthesis. URL: SD-111-00157 |
| Market Analysis Reports |