Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Highpharm Medchem (Ningbo) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.highpharm.com | |||
 | +86 (574) 5566-3958 | |||
 | +86 (574) 5566-3960 | |||
 | highpharm@highpharm.com lisayang925@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Shanghai Sinofluoro chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinofluoro.com | |||
 | +86 (21) 6427-9360 | |||
 | +86 (21) 6427-8603 | |||
 | simon.lv@sinofluoro.com jerry.chen@sinofluoro.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| LinkChem Shanghai Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.linkchem.cn | |||
 | +86 (21) 5895-0110 +86 13391192982 | |||
 | +86 (21) 2022-2982 | |||
 | sales@linkchem.cn | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Shanghai Sinoflychem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinoflychem.com | |||
 | +86 (21) 3530-3826 | |||
 | +86 (21) 3530-0619 | |||
 | johnson@sinoflychem.com shenxiangchemphar@gmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Taizhou Tongxin Biopharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.allyrise.com | |||
 | +86 18652728585 | |||
 | +86 (523) 8276-5215 | |||
 | sales@allyrise.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Coresyn Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.coresyn.com | |||
 | +86 (571) 8662-6709 | |||
 | sales@coresyn.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Witofly Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.witofly.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales@witofly.com | |||
 | QQ Chat | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Daji Medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.djpharm.com | |||
 | +86 13816245196 | |||
 | sales1@djpharm.com | |||
| Chemical distributor since 2019 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Combi-Blocks, Inc. | USA | |||
|---|---|---|---|---|
 | www.combi-blocks.com | |||
 | +1 (858) 635-8950 | |||
 | +1 (858) 635-8991 | |||
 | sales@combi-blocks.com | |||
| Chemical manufacturer | ||||
| SynChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.synchem.com | |||
 | +1 (847) 298-2436 ex 230 | |||
 | +1 (847) 298-2439 | |||
 | sales@synchem.com | |||
| Chemical manufacturer since 1997 | ||||
| Synthonix, Inc. | USA | |||
|---|---|---|---|---|
 | www.synthonix.com | |||
 | +1 (919) 875-9277 | |||
 | +1 (919) 875-9601 | |||
 | info@synthonix.com | |||
| Chemical manufacturer | ||||
| Activate Scientific GmbH | Germany | |||
|---|---|---|---|---|
 | www.activate-scientific.com | |||
 | +49 (8051) 965-1660 | |||
 | +49 (8051) 965-1661 | |||
 | rschmitt@activate-scientific.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Imidazoles |
|---|---|
| Name | 1-Tritylimidazole-4-carboxaldehyde |
| Synonyms | 1-Trityl-1H-imidazole-4-carboxaldehyde |
| Molecular Structure | 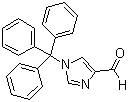 |
| Molecular Formula | C23H18N2O |
| Molecular Weight | 338.40 |
| CAS Registry Number | 33016-47-6 |
| EC Number | 620-447-0 |
| SMILES | C1=CC=C(C=C1)C(C2=CC=CC=C2)(C3=CC=CC=C3)N4C=C(N=C4)C=O |
| Density | 1.1±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 192 °C (Decomposes) (Expl.) |
| Boiling point | 502.8±45.0 °C 760 mmHg (Calc.)* |
| Flash point | 257.9±28.7 °C (Calc.)* |
| Index of refraction | 1.613 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
1-Tritylimidazole-4-carboxaldehyde is a heterocyclic compound in which an imidazole ring is substituted with a trityl group at the nitrogen atom in position 1 and a formyl group at position 4. Its molecular formula is C26H21N2O, with a molecular weight of approximately 365.46 g/mol. The trityl group is a bulky triphenylmethyl substituent that provides steric protection to the nitrogen, while the aldehyde at position 4 introduces an electrophilic site, making the molecule versatile for further functionalization. The synthesis of 1-tritylimidazole-4-carboxaldehyde generally involves protection of the imidazole nitrogen with a trityl chloride or trityl bromide under basic conditions. The trityl group serves as a protecting group to prevent undesired reactions at the nitrogen during subsequent transformations. The formyl group at position 4 can be introduced through a Vilsmeier–Haack reaction or other selective formylation methods that target the activated position of the imidazole ring. Reaction conditions are carefully controlled to avoid overformylation or decomposition of the trityl-protected imidazole. Chemically, the aldehyde functionality at position 4 is highly reactive and can participate in nucleophilic addition reactions, including formation of imines, hydrazones, and alcohols upon reduction. The trityl group on the nitrogen reduces the basicity and nucleophilicity of the imidazole, stabilizing the compound under various reaction conditions and allowing selective transformations at the aldehyde. Deprotection of the trityl group can be performed under acidic conditions to restore the free imidazole nitrogen for further reactions. The compound is typically a solid at room temperature and shows good solubility in nonpolar and moderately polar organic solvents such as dichloromethane, chloroform, and tetrahydrofuran. Its stability under ambient conditions is adequate for routine handling, though care should be taken to avoid strong acids or oxidizing agents that could compromise the aldehyde or induce cleavage of the trityl group. The bulky trityl substituent also influences the steric environment of the molecule, affecting reactivity and interactions in both synthetic and crystallization processes. In practical applications, 1-tritylimidazole-4-carboxaldehyde is primarily used as an intermediate in the synthesis of functionalized imidazole derivatives. The aldehyde group provides a site for further derivatization, while the trityl group allows for temporary protection of the nitrogen during multi-step synthetic sequences. This combination is particularly valuable in medicinal chemistry and heterocyclic synthesis, where selective modification of imidazole derivatives is necessary for preparing bioactive compounds, ligands, or functional materials. Overall, 1-tritylimidazole-4-carboxaldehyde combines a sterically protected imidazole core with a reactive aldehyde functionality, offering a versatile building block for organic synthesis. Its structural features support selective functionalization and controlled transformations, making it useful for the development of complex heterocyclic molecules and functionalized intermediates. References 2017. A novel and facile route for the synthesis of medetomidine as the α2-adrenoceptor agonist. Journal of the Iranian Chemical Society, 14(6). DOI: 10.1007/s13738-017-1114-0 2014. Fluorinated Imidazoles and Benzimidazoles. Fluorine in Heterocyclic Chemistry Volume 1. DOI: 10.1007/978-3-319-04346-3_8 |
| Market Analysis Reports |