Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Nanjing Finetech Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fine-chemtech.com | |||
 | +86 (25) 5207-8417 +86 17714198479 | |||
 | +86 (25) 5207-8417 | |||
 | sales@fine-chemtech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Zhejiang Ausun Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ausunpharm.com | |||
 | +86 (576) 8558-9335 +86 13957690608 | |||
 | +86 (576) 8558-9168 | |||
 | sales@ausunpharm.com | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Taizhou KEDE Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kedechemical.com | |||
 | +86 (576) 8461-3060 +86 13093829633 | |||
 | +86 (576) 8461-3060 | |||
 | sales@kedechemical.com | |||
 | QQ Chat | |||
 | WeChat: 13093829633 | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Nanjing Xize Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.njxizebio.com | |||
 | +86 (25) 6602-3220 | |||
 | +86 (25) 6602-3220 | |||
 | sale@njxizebio.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Genriver Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.genriverpharm.com | |||
 | +86 13761582449 +86 13482015261 | |||
 | +86 (21) 3778-2903 | |||
 | info@genriverpharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Taizhou Runsun Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.runsunchemical.com | |||
 | +86 15867635987 | |||
 | sales@runsunchemical.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Pure Bio Medical Jiangsu Ltd. | China | |||
|---|---|---|---|---|
 | www.purejs.cn | |||
 | +86 (025) 8560-7383 | |||
 | jiangziwei@purejs.cn | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | Biochemical >> Inhibitor >> Transmembrane transporters >> CFTR activator |
|---|---|
| Name | Lubiprostone |
| Synonyms | (2R,4aR,5R,7aR)-2-(1,1-Difluoropentyl)-2-hydroxy-6-oxo-3,4,4a,5,7,7a-hexahydrocyclopenta[b]pyran-5-heptanoic acid |
| Molecular Structure | 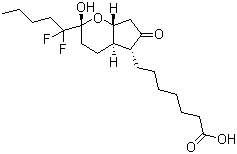 |
| Molecular Formula | C20H32F2O5 |
| Molecular Weight | 390.46 |
| CAS Registry Number | 333963-40-9 |
| EC Number | 603-977-7 |
| SMILES | CCCCC([C@]1(CC[C@H]2[C@H](O1)CC(=O)[C@@H]2CCCCCCC(=O)O)O)(F)F |
| Density | 1.2±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 532.3±50.0 °C 760 mmHg (Calc.)* |
| Flash point | 275.7±30.1 °C (Calc.)* |
| Index of refraction | 1.486 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P280-P301+P312-P302+P352-P305+P351+P338 Details |
| SDS | Available |
|
Lubiprostone is a synthetic bicyclic fatty acid derivative classified as a prostaglandin E1 (PGE1) analog, developed to treat chronic constipation, irritable bowel syndrome with constipation (IBS-C), and opioid-induced constipation in adults. It was discovered as part of research into prostaglandin derivatives capable of modulating ion transport in epithelial cells without producing the systemic effects associated with native prostaglandins. Its mechanism of action involves the selective activation of type 2 chloride channels (ClC-2) in the apical membrane of gastrointestinal epithelial cells, which promotes chloride ion secretion into the intestinal lumen. This chloride movement osmotically draws water into the lumen, softening stool and increasing intestinal motility. The discovery of lubiprostone followed structure–activity relationship studies of prostaglandin E1 analogs aimed at enhancing local gastrointestinal activity while minimizing systemic absorption. Lubiprostone’s chemical design includes a bicyclic structure with hydroxyl groups strategically positioned to maintain chloride channel activity and selectivity. Pharmacokinetic studies show minimal systemic exposure following oral administration, as the compound acts locally in the gastrointestinal tract. Its metabolism is primarily via oxidation and reduction in the stomach and intestine, and the drug is excreted mostly in feces. Clinical development included preclinical studies demonstrating chloride secretion in epithelial cell models and animal models of constipation. Electrophysiological studies confirmed that lubiprostone stimulates chloride currents independently of cyclic AMP pathways, differentiating it mechanistically from other secretagogues that act through CFTR channels. These findings supported its use as a locally acting intestinal secretagogue without significant cardiovascular or systemic prostaglandin effects. Randomized, double-blind, placebo-controlled clinical trials confirmed lubiprostone’s efficacy in improving spontaneous bowel movement frequency, stool consistency, and patient-reported outcomes. In patients with chronic idiopathic constipation, lubiprostone increased the number of complete spontaneous bowel movements per week, reduced straining, and alleviated abdominal discomfort. Similar studies in IBS-C demonstrated symptom improvement in women, including relief from bloating and abdominal pain. For opioid-induced constipation, lubiprostone also improved bowel habits in patients receiving chronic opioid therapy without altering opioid analgesia. Lubiprostone is generally well tolerated, with the most common adverse effects being nausea and diarrhea. Other less frequent effects include headache and abdominal distension. Its localized mechanism of action contributes to a favorable safety profile compared with systemic prostaglandin analogs. Lubiprostone’s development established a precedent for the design of prostaglandin derivatives that target epithelial ion channels rather than classical prostanoid receptors, expanding therapeutic options for constipation-related disorders. References Cuppoletti J, Malinowska DH, Tewari KP, Li QJ, Sherry AM, Patchen ML, Ueno R (2004) SPI-0211 activates T84 cell chloride transport and recombinant human ClC-2 chloride currents. American Journal of Physiology - Cell Physiology 287 C1173–C1183 DOI: 10.1152/ajpcell.00528.2003 Camilleri M, Bharucha AE, Ueno R, Burton D, Thomforde GM, Baxter K, McKinzie S, Zinsmeister AR (2006) Effect of a selective chloride channel activator, lubiprostone, on gastrointestinal transit, gastric sensory, and motor functions in healthy volunteers. American Journal of Physiology - Gastrointestinal and Liver Physiology 290 G942–G947 DOI: 10.1152/ajpgi.00264.2005 Bassil AK, Borman RA, Jarvie EM, McArthur-Wilson RJ, Weston J, Lee K, Sanger GJ (2008) Activation of prostaglandin EP receptors by lubiprostone in rat and human stomach and colon. British Journal of Pharmacology 154 126–135 DOI: 10.1038/bjp.2008.84 |
| Market Analysis Reports |