Online Database of Chemicals from Around the World
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Sphinx Scientific Laboratory (Tianjin) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sphinxpharm.com | |||
 | +86 (22) 6621-1289 ex 818 | |||
 | +86 (22) 6621-1370 | |||
 | lilh@sphinxpharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Finetech Industry Limited | China | |||
|---|---|---|---|---|
 | www.finetechchem.com | |||
 | +86 (27) 8746-5837 +86 18971612321 | |||
 | +86 (27) 8777-2287 | |||
 | sales@finetechnology-ind.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2019 | ||||
| ABX Advanced Biochemical Compounds | Germany | |||
|---|---|---|---|---|
 | www.abx.de | |||
 | +49 (3528) 4041-60 | |||
 | +49 (3528) 4041-65 | |||
 | info@abx.de | |||
| Chemical manufacturer | ||||
| Linhai Wonderful-Trading Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.wonderfult.com | |||
 | +86 (576) 8568-6581 8568-6706 | |||
 | +86 (576) 8568-6328 | |||
 | sales@wonderfult.com | |||
| Chemical distributor | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | API >> Circulatory system medication >> Anti-shock vasoactive drug |
|---|---|
| Name | Metaraminol bitartrate |
| Synonyms | (-)-m-Hydroxyphenylpropanolamine bitartrate salt |
| Molecular Structure | 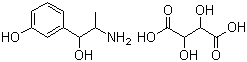 |
| Molecular Formula | C9H13NO2.C4H6O6 |
| Molecular Weight | 317.29 |
| CAS Registry Number | 33402-03-8 |
| EC Number | 251-502-3 |
| SMILES | C[C@@H]([C@@H](C1=CC(=CC=C1)O)O)N.[C@@H]([C@H](C(=O)O)O)(C(=O)O)O |
| Solubility | 10 mM (water) |
|---|---|
| Hazard Symbols |  GHS06 Danger Details GHS06 Danger Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H311-H331 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P270-P271-P280-P302+P352-P304+P340-P310-P330-P361-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| Transport Information | UN 2811 | ||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Metaraminol bitartrate, a sympathomimetic amine, was discovered in the mid-20th century. It was synthesized as a derivative of phenylethylamine, aiming to develop a compound with potent vasoconstrictive properties. The compound's ability to stimulate alpha-adrenergic receptors and increase peripheral vascular resistance was found to be particularly useful in the management of hypotension, especially in surgical and emergency settings. Its discovery marked a significant advancement in pharmacology, providing clinicians with a valuable tool for addressing acute episodes of low blood pressure. Metaraminol bitartrate is primarily used for the treatment of hypotension, a condition characterized by abnormally low blood pressure. By stimulating alpha-adrenergic receptors in blood vessels, it causes vasoconstriction, leading to increased blood pressure. This effect is particularly beneficial in scenarios such as hemorrhagic shock, septic shock, and anesthesia-induced hypotension, where rapid restoration of blood pressure is crucial for patient stability. In anesthesia practice, metaraminol bitartrate is frequently administered to counteract hypotension induced by anesthetic agents or physiological changes during surgery. Its ability to rapidly elevate blood pressure makes it an essential adjunct during anesthesia induction and maintenance, ensuring adequate tissue perfusion and oxygen delivery to vital organs. Metaraminol bitartrate plays a critical role in emergency medicine, where the prompt correction of hypotension is vital for patient survival. It is commonly used in resuscitation efforts for patients in shock, cardiac arrest, or other life-threatening conditions. The drug's rapid onset of action and potent vasoconstrictive effects make it an invaluable tool in stabilizing hemodynamics and improving tissue perfusion in critically ill patients. In obstetric practice, metaraminol bitartrate may be employed to manage hypotension associated with spinal anesthesia during cesarean section or other obstetric procedures. By increasing systemic vascular resistance, it helps maintain maternal blood pressure within the optimal range, reducing the risk of maternal and fetal complications during delivery. Beyond clinical applications, metaraminol bitartrate continues to be studied for its pharmacological properties and potential therapeutic uses. Research efforts focus on optimizing dosing regimens, exploring novel delivery methods, and investigating its efficacy in specific patient populations. These studies aim to further enhance the drug's effectiveness and safety in various clinical settings. References 2017. A randomised double-blind trial of phenylephrine and metaraminol infusions for prevention of hypotension during spinal and combined spinal-epidural anaesthesia for elective caesarean section. Anaesthesia, 72(5). DOI: 10.1111/anae.13836 2019. Metaraminol use during spinal anaesthesia for caesarean section: a meta-analysis of randomised controlled trials. International Journal of Obstetric Anesthesia, 39. DOI: 10.1016/j.ijoa.2019.01.009 2021. Pharmacoepidemiology of metaraminol in critically ill patients with shock in a tertiary care hospital. Australian critical care : official journal of the Confederation of Australian Critical Care Nurses, 34(6). DOI: 10.1016/j.aucc.2021.01.002 |
| Market Analysis Reports |