Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Classification | Chemical reagent >> Organic reagent >> Ether |
|---|---|
| Name | 1,2-Bis(2-cyanoethoxy)ethane |
| Synonyms | 3-[2-(2-cyanoethoxy)ethoxy]propanenitrile |
| Molecular Structure | 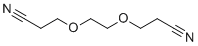 |
| Molecular Formula | C8H12N2O2 |
| Molecular Weight | 168.19 |
| CAS Registry Number | 3386-87-6 |
| EC Number | 222-208-2 |
| SMILES | C(COCCOCCC#N)C#N |
| Solubility | 3.476e+005 mg/L (25 °C water) |
|---|---|
| Density | 1.1±0.1 g/cm3, Calc.* |
| Index of Refraction | 1.443, Calc.* |
| Melting point | 73.92 °C |
| Boiling Point | 308.79 °C, 358.5±22.0 °C (760 mmHg), Calc.* |
| Flash Point | 154.0±15.2 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS06;GHS07 Danger Details GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H315-H319 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P501-P270-P264-P280-P302+P352-P337+P313-P305+P351+P338-P362+P364-P332+P313-P301+P310+P330-P405 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
1,2-Bis(2-cyanoethoxy)ethane is a chemical compound that features an ethane backbone with two 2-cyanoethoxy groups attached at the 1 and 2 positions. This compound is characterized by its structural configuration, which includes both cyano and ethoxy functionalities. The presence of the cyano group imparts reactivity to the molecule, while the ethoxy groups contribute to its solubility and stability in organic solvents. The compound has been widely studied in the context of organic synthesis and materials science. It is typically used as a reagent or intermediate in various chemical reactions, particularly those involving nucleophilic substitution due to the electron-withdrawing nature of the cyano group. The presence of both cyano and ethoxy groups makes it a useful building block for the construction of more complex molecular structures. In synthetic chemistry, 1,2-bis(2-cyanoethoxy)ethane has been employed in the preparation of polymeric materials. The cyano group is particularly useful for introducing specific reactivity, which can be exploited in the synthesis of polymers with specialized properties. For instance, the compound can participate in polymerization reactions or be used as a monomer in the formation of copolymers, contributing to materials that possess unique mechanical, electrical, or thermal properties. The ethoxy groups in the compound also enhance its solubility in organic solvents, making it a favorable candidate in applications where solubility in non-polar or moderately polar solvents is important. Additionally, the combination of cyano and ethoxy groups in its structure contributes to its stability under various chemical conditions, further expanding its utility in different chemical processes. In the field of material science, 1,2-bis(2-cyanoethoxy)ethane has been investigated for its potential use in the development of functional materials, including those for electronic applications. The cyano group is known for its ability to influence the electronic properties of materials, which is valuable in the design of materials with specific conductivity or reactivity. As a result, this compound has been explored as part of electronic devices, sensors, and coatings. Overall, 1,2-bis(2-cyanoethoxy)ethane is a versatile chemical intermediate with applications in organic synthesis, materials science, and the development of functionalized polymers. Its structural features, including the presence of cyano and ethoxy groups, allow it to play a role in a variety of chemical reactions and material formulations. References 2001. Reaction of 4,4-Dimethyl-1,3-dioxane with Dinitriles. Russian Journal of General Chemistry, 71(1). DOI: 10.1023/a:1012302127645 2014. Dinitrile compound containing ethylene oxide moiety with enhanced solubility of lithium salts as electrolyte solvent for high-voltage lithium-ion batteries. Ionics, 21(4). DOI: 10.1007/s11581-014-1272-3 2022. An acrylate-based quasi-solid polymer electrolyte incorporating a novel dinitrile poly(ethylene glycol) plasticizer for lithium-ion batteries. Journal of Materials Science, 57(28). DOI: 10.1007/s10853-022-07431-1 |
| Market Analysis Reports |