Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Changzhou Carbochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.carbo-chem.com | |||
 | +86 (519) 8918-1862 +86 13775204319 | |||
 | +86 (519) 8918-1862 | |||
 | gao@carbo-chem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Qingdao Kingway Pharmtech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingwaypharm.com | |||
 | +86 (532) 8711-8899 | |||
 | +86 (532) 8411-7788 | |||
 | sales@kingwaypharm.com kingwaypharm@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Xuchang Chenhe Bio-pharmaceutical Tech. Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.the-chem.com | |||
 | +86 (374) 579-3128 +86 15836530076 | |||
 | +86 (374) 579-3128 | |||
 | wxh5168@126.com wxh5168@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Qichuang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qc-chemical.com | |||
 | +86 (571) 8893-5129 | |||
 | +86 (571) 8825-0182 | |||
 | david@qc-chemical.com sales@qc-chemical.com davidw0828@gmail.com | |||
 | QQ Chat | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Shanghai Neosun Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neosunpharm.com | |||
 | +86 (21) 2095-6531 +86 18116226605 +86 18939712385 | |||
 | +86 (21) 2095-6532 | |||
 | neosunpharm@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Suzhou Bichal Biological Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bichal.com | |||
 | +86 (512) 6805-1130 | |||
 | +86 (512) 6805-2322 | |||
 | info@bichal.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Sanochem (Chengdu) Tech. Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sano-chem.com | |||
 | +86 15002851260 | |||
 | +86 (28) 8799-9436 | |||
 | sales@sano-chem.com | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Rochi Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rochipharma.com | |||
 | +86 (21) 3875-1876 +86 15000076078 | |||
 | +86 (21) 5027-5764 | |||
 | info@rochipharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Cherry Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cherrypharmatech.com | |||
 | +86 (571) 8163-6070 +86 18042403330 | |||
 | +86 (571) 8163-6070 | |||
 | info@cherrypharmatech.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Coresyn Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.coresyn.com | |||
 | +86 (571) 8662-6709 | |||
 | sales@coresyn.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Edu Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemido.com | |||
 | +86 19301251526 | |||
 | sales1@chemido.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Organic raw materials >> Amino compound >> Amide compound |
|---|---|
| Name | 6-(4-Methylpiperazin-1-yl)-4-(2-methylphenyl)nicotinamide |
| Synonyms | 4-(2-Methylphenyl)-6-(4-methyl-1-piperazinyl)-3-pyridinecarboxamide |
| Molecular Structure | 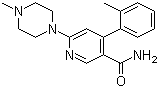 |
| Molecular Formula | C18H22N4O |
| Molecular Weight | 310.39 |
| CAS Registry Number | 342417-01-0 |
| SMILES | CC1=CC=CC=C1C2=CC(=NC=C2C(=O)N)N3CCN(CC3)C |
| Density | 1.2±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 164-165 °C (Expl.) |
| Boiling point | 498.1±45.0 °C 760 mmHg (Calc.)* |
| Flash point | 255.0±28.7 °C (Calc.)* |
| Index of refraction | 1.601 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302 Details |
| Safety Statements | P280-P305+P351+P338 Details |
| SDS | Available |
|
6-(4-Methylpiperazin-1-yl)-4-(2-methylphenyl)nicotinamide is a heteroaromatic amide in which a nicotinamide core is substituted at position 4 with a 2-methylphenyl group and at position 6 with a 4-methylpiperazin-1-yl group. Its molecular formula is C18H22N4O, with a molecular weight of approximately 306.40 g/mol. The pyridine ring of nicotinamide provides aromaticity and electronic delocalization, while the amide functionality and piperazine substituent introduce hydrogen-bonding capacity, polarity, and sites for further chemical modifications. The compound is generally synthesized through selective functionalization of a nicotinamide precursor. The 4-position aryl substitution can be achieved via palladium-catalyzed cross-coupling reactions, such as Suzuki or Buchwald–Hartwig couplings, with appropriately substituted halonicotinamides and 2-methylphenyl boronic acid or amine derivatives. Introduction of the 6-(4-methylpiperazin-1-yl) group typically involves nucleophilic substitution of a leaving group on the nicotinamide ring by the secondary amine of 4-methylpiperazine under basic conditions. Reaction optimization ensures regioselectivity and minimizes side reactions, preserving the integrity of the pyridine and amide functionalities. Chemically, the amide group provides a site for hydrogen bonding and can undergo typical reactions such as acylation, amidation, or formation of imides under controlled conditions. The piperazine nitrogen atoms can act as nucleophiles, bases, or protonation sites, influencing solubility, polarity, and binding interactions in chemical and biological contexts. The 2-methylphenyl group contributes steric bulk and hydrophobic character, which can modulate intermolecular interactions and influence reactivity at adjacent positions on the nicotinamide ring. 6-(4-Methylpiperazin-1-yl)-4-(2-methylphenyl)nicotinamide is generally a solid at room temperature and demonstrates solubility in polar organic solvents such as ethanol, dimethylformamide, and dichloromethane. Its stability is adequate under ambient conditions, although strong acids, bases, or oxidizing agents should be avoided to prevent amide hydrolysis or degradation of the piperazine ring. The combination of polar and nonpolar substituents affects crystallization, solubility, and potential intermolecular interactions in both synthetic and application contexts. In practical applications, this compound is primarily used as a synthetic intermediate in medicinal chemistry. The combination of a nicotinamide core, an aryl group, and a piperazine substituent enables structural diversity, providing a platform for the preparation of bioactive derivatives, receptor ligands, or heterocyclic scaffolds. Its functional groups allow selective chemical modification, facilitating exploration of structure–activity relationships and optimization of physicochemical properties in drug discovery and chemical biology. Overall, 6-(4-methylpiperazin-1-yl)-4-(2-methylphenyl)nicotinamide combines a heteroaromatic core with reactive amide and amine functionalities and a sterically defined aryl substituent. Its versatile chemical behavior and defined structural features make it a valuable intermediate for the synthesis of complex molecules with potential biological or chemical applications. References 2016. Netupitant. Pharmaceutical Substances. URL: KD-14-0114 |
| Market Analysis Reports |