Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Epochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.epochem.com | |||
 | +86 (21) 6760-1595 6760-1597 | |||
 | +86 (21) 6760-1605 | |||
 | seth_wang@epochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Kingfirst Chemical (Nanjing) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingfirstchem.com | |||
 | +86 (25) 8452-2992 8477-3604 | |||
 | +86 (25) 8452-1617 | |||
 | marketing@kingfirstchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| HBCChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.hbcchem-inc.com | |||
 | +1 (510) 219-6317 | |||
 | +1 (510) 675-0318 | |||
 | sales@hbcchem-inc.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| SynQuest Labs, Inc. | USA | |||
|---|---|---|---|---|
 | www.synquestlabs.com | |||
 | +1 (386) 462-0788 | |||
 | +1 (386) 462-7097 | |||
 | Sales@synquestlabs.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Beijing Infoark Technology Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.infoark.com.cn | |||
 | +86 (10) 5166-8333 ex 221, 240, 211 | |||
 | +86 (10) 8815-2455 ex 221, 240, 211 | |||
 | intermediate@infoark.com.cn yanlixie_cn@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Boroncore LLC | China | |||
|---|---|---|---|---|
 | www.boroncore.com | |||
 | +86 (571) 8558-6753 | |||
 | custom@boroncore.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Zhangjiagang Clent Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.clentchem.com | |||
 | +86 (512) 5881-3106 +86 13962460297 | |||
 | +86 (512) 5881-2106 | |||
 | sales@clentchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Ningbo NFC Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.nfcchem.com | |||
 | +86 (574) 8799-1182 | |||
 | +86 (574) 8799-1185 | |||
 | sales@nfcchem.com | |||
 | QQ Chat | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Nanjing Duolong Bio-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.dolonchem.com | |||
 | +86 (25) 8326-3139 | |||
 | +86 (25) 8326-3139 | |||
 | sales@dolonchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2014 | ||||
| NetQem, LLC | USA | |||
|---|---|---|---|---|
 | www.netqem.us | |||
 | +1 (919) 544-4122 | |||
 | +1 (919) 544-4109 | |||
 | sales@netqem.us | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Smartchem (Beijing) Ltd. | China | |||
|---|---|---|---|---|
 | www.smartchembeijing.com | |||
 | +86 (10) 5720-3829 5130-5129 +86 13301124250 | |||
 | +86 (10) 5130-5388 | |||
 | sales@smartchembeijing.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Wuxi Qianhao Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bestpharm.cn | |||
 | +86 (510) 8641-9776 | |||
 | +86 (21) 6117-9676 | |||
 | sales@bestpharm.cn | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Wonderchem Co., Limited | Hong Kong | |||
|---|---|---|---|---|
 | www.wonder-chem.com | |||
 | +852 8192-5218 | |||
 | sales@wonder-chem.com | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Hangzhou Hairui Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hairuichem.com | |||
 | +86 (571) 8669-1155 | |||
 | +86 (571) 8669-1154 | |||
 | sales@hairuichem.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2017 | ||||
| R&D Scientific Inc. | Canada | |||
|---|---|---|---|---|
 | www.rdscientific.com | |||
 | +1 (226) 600-0236 | |||
 | sales@rdscientific.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Auben Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.aubenchem.com | |||
 | +86 19121901603 | |||
 | sales@aubenchem.com | |||
| Chemical distributor since 2023 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Carbone Scientific Co., Ltd. | UK | |||
|---|---|---|---|---|
 | www.carbonesci.com | |||
 | +44 (870) 486-8629 | |||
 | +44 (870) 288-7399 | |||
 | sales@carbonesci.com | |||
| Chemical distributor | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| AK Scientific, Inc | USA | |||
|---|---|---|---|---|
 | www.aksci.com | |||
 | +1 (510) 429-8835 | |||
 | +1 (510) 429-8836 | |||
 | sales@aksci.com | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Anisyn, Inc. | USA | |||
|---|---|---|---|---|
 | www.anisyn.com | |||
 | +1 (269) 372-8736 | |||
 | +1 (269) 372-3397 | |||
 | sales@anisyn.com | |||
| Chemical manufacturer | ||||
| Classification | API >> Synthetic anti-infective drugs >> Sulfonamides and synergists |
|---|---|
| Name | (S)-tert-Butylsulfinamide |
| Synonyms | (S)-(-)-2-Methyl-2-propanesulfinamide |
| Molecular Structure | 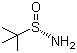 |
| Molecular Formula | C3H11CNOS |
| Molecular Weight | 121.20 |
| CAS Registry Number | 343338-28-3 |
| EC Number | 640-158-3 |
| SMILES | CC(C)(C)[S@](=O)N |
| Density | 1.1±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 97 - 101 °C (Expl.) |
| Boiling point | 220.0±23.0 °C 760 mmHg (Calc.)* |
| Flash point | 86.8±22.6 °C (Calc.)* |
| Index of refraction | 1.523 (Calc.)* |
| Alpha | -4.5 ° (c=1, CHCl3) (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
(S)-tert-Butylsulfinamide is an organosulfur compound consisting of a tert-butyl group attached to a sulfinamide functional group, with defined (S)-stereochemistry at the sulfur atom. Its molecular formula is C4H11NOS. The compound appears as a colorless crystalline solid and is soluble in polar organic solvents such as dichloromethane, tetrahydrofuran, and ethanol, while exhibiting limited solubility in water. The chiral sulfinamide moiety provides both nucleophilic and electrophilic reactivity, making it a versatile building block in asymmetric synthesis. The discovery of (S)-tert-butylsulfinamide is tied to the development of chiral auxiliaries and chiral amine synthesis. Sulfinamides were recognized for their ability to induce stereocontrol in nucleophilic addition reactions due to the stereogenic sulfur atom. The tert-butyl group imparts steric bulk that enhances diastereoselectivity during reactions with carbonyl compounds, imines, and electrophiles. As a result, (S)-tert-butylsulfinamide is widely used as a chiral auxiliary to produce enantiomerically enriched amines and other nitrogen-containing compounds. Synthesis of (S)-tert-butylsulfinamide typically involves oxidation of tert-butylthiol or tert-butylsulfide derivatives to generate the corresponding sulfinyl chloride, followed by reaction with ammonia or an amine source under controlled conditions to form the sulfinamide. Enantioselective oxidation or resolution strategies are employed to obtain the (S)-enantiomer with high optical purity. Purification is generally accomplished by recrystallization from suitable organic solvents, yielding a stable, stereochemically defined product. Chemically, (S)-tert-butylsulfinamide exhibits nucleophilic behavior at the nitrogen atom and electrophilic behavior at the sulfur center. The sulfur atom is stereogenic, allowing it to direct stereoselective reactions, particularly in the formation of imines and subsequent nucleophilic additions. The tert-butyl group shields one face of the sulfinamide, enabling high diastereoselectivity when reacting with prochiral electrophiles. The compound is stable under ambient conditions but can undergo oxidation to the corresponding sulfonamide or reduction to the corresponding thiol under appropriate conditions. In organic synthesis, (S)-tert-butylsulfinamide is primarily employed as a chiral auxiliary in the asymmetric synthesis of amines, including α-amino acids, alkaloids, and pharmaceutical intermediates. It can form N-sulfinyl imines with aldehydes or ketones, which then undergo stereoselective nucleophilic addition to produce enantiomerically enriched amines after deprotection. This strategy allows precise control over absolute configuration and has been applied in the synthesis of complex molecules for medicinal chemistry. From a physical perspective, the compound is thermally stable and can be handled under normal laboratory conditions. It is typically stored in tightly sealed containers to avoid moisture, which may affect its crystalline structure or lead to hydrolysis. Standard laboratory safety procedures, including gloves and eye protection, should be followed when handling, as it is an irritant to skin and eyes. Overall, (S)-tert-butylsulfinamide is a chiral organosulfur compound of significant importance in asymmetric synthesis. Its stereogenic sulfur atom, combined with the steric influence of the tert-butyl group, enables the production of enantiomerically enriched amines with high selectivity. The compound serves as a cornerstone reagent in the synthesis of pharmaceuticals, natural products, and complex nitrogen-containing molecules, exemplifying the utility of chiral sulfinamides in modern organic chemistry. References 2024. A comprehensive review on synthetic strategy and MOA of marketed drugs having therapeutically potential chemical entity pyrazole. Journal of the Iranian Chemical Society. DOI: 10.1007/s13738-024-03095-7 |
| Market Analysis Reports |