Online Database of Chemicals from Around the World
| SINOJIE (HK) Limited | China | |||
|---|---|---|---|---|
 | www.sinojie.com | |||
 | +86 (21) 5230-3877 | |||
 | +86 (21) 5230-3799 | |||
 | sissizhou@sinojie.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Arasan Chemicals Private Limited | India | |||
|---|---|---|---|---|
 | www.arasanchemicals.com | |||
 | +91 (98) 2001-1049 | |||
 | +91 (22) 6615-3292 | |||
 | contact@arasanchemicals.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Intatrade Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.intatrade.de | |||
 | +49 (3493) 605-465 | |||
 | +49 (3493) 605-470 | |||
 | sales@intatrade.de | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Zhangjiagang Clent Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.clentchem.com | |||
 | +86 (512) 5881-3106 +86 13962460297 | |||
 | +86 (512) 5881-2106 | |||
 | sales@clentchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Zentra Bio-Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zentrachem.com | |||
 | 18321999949 | |||
 | steven@zentrachem.com | |||
 | QQ Chat | |||
 | WeChat: Hangzhou Zentra Bio-Chemical Co., Ltd. | |||
| Chemical distributor since 2025 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Changzhou Ruiming Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chengnanchem.com | |||
 | +86 (519) 8273-1357 | |||
 | +86 (519) 8273-1720 | |||
 | swm@chengnanchem.com | |||
| Chemical manufacturer | ||||
| Classification | Food additive >> Preservative |
|---|---|
| Name | 2'-Hydroxy-3-phenylpropiophenone |
| Synonyms | 1-(2-Hydroxyphenyl)-3-phenylpropan-1-one |
| Molecular Structure | 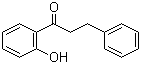 |
| Molecular Formula | C15H14O2 |
| Molecular Weight | 226.27 |
| CAS Registry Number | 3516-95-8 |
| EC Number | 222-521-4 |
| SMILES | C1=CC=C(C=C1)CCC(=O)C2=CC=CC=C2O |
| Density | 1.2±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 36 - 37 °C (Expl.) |
| Boiling point | 381.1±25.0 °C 760 mmHg (Calc.)* |
| Flash point | 162.7±15.8 °C (Calc.)*, 113 °C (Expl.) |
| Index of refraction | 1.6 (Calc.)*, 1.5968 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS09 Warning Details GHS07;GHS09 Warning Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335-H400-H410 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P273-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
2’-Hydroxy-3-phenylpropiophenone is an organic compound characterized by a propiophenone core substituted with a hydroxyl group at the 2’-position and a phenyl group at the 3-position on the aromatic ring. The propiophenone structure consists of a benzene ring attached to a three-carbon ketone side chain (propiophenone), where the carbonyl group is positioned adjacent to the benzene ring. The hydroxyl substitution at the 2’-position refers to the ortho position relative to the ketone side chain, while the phenyl substituent at the 3-position occupies the meta position relative to the ketone. The presence of both hydroxyl and phenyl substituents on the aromatic ring influences the compound’s chemical reactivity and physical properties. The hydroxyl group provides a site for hydrogen bonding and can participate in electrophilic and nucleophilic reactions. The additional phenyl ring contributes to increased hydrophobicity and may affect molecular interactions. Synthesis of 2’-hydroxy-3-phenylpropiophenone can be achieved via Friedel–Crafts acylation reactions where a hydroxy-substituted benzene derivative undergoes acylation with phenylpropionyl chloride or related reagents, or through alternative carbonylation methods involving phenyl precursors and appropriate catalysts. Selective substitution patterns are crucial during synthesis to ensure the hydroxyl and phenyl groups are positioned at the correct sites on the aromatic ring. Chemically, the compound exhibits typical properties of hydroxyaryl ketones, including potential keto-enol tautomerism, nucleophilic addition at the carbonyl carbon, and electrophilic aromatic substitution reactions facilitated by the activating hydroxyl group. The hydroxyl group’s ortho position relative to the carbonyl also allows for intramolecular hydrogen bonding, which may influence the compound’s conformation and stability. Applications of 2’-hydroxy-3-phenylpropiophenone are mainly in organic synthesis and as intermediates in the preparation of pharmaceuticals, agrochemicals, and fine chemicals. The compound’s reactive sites enable it to serve as a versatile building block for constructing more complex molecular architectures. Its phenolic and ketone functionalities offer opportunities for further chemical modifications such as esterification, etherification, and condensation reactions. Analytical characterization of 2’-hydroxy-3-phenylpropiophenone commonly involves nuclear magnetic resonance (²H and ³C NMR) spectroscopy to identify the aromatic proton environments, hydroxyl proton signals, and carbonyl carbon. Infrared (IR) spectroscopy detects characteristic absorption bands of hydroxyl groups (around 3400 cm−1) and ketone carbonyl stretches (near 1700 cm−1). Mass spectrometry confirms molecular weight and fragmentation patterns consistent with the substituted propiophenone structure. Physically, the compound typically exists as a solid with moderate solubility in organic solvents such as ethanol, methanol, and dichloromethane, but limited solubility in water due to its hydrophobic aromatic rings. Its melting point and crystallinity depend on purity and specific isomeric form. In summary, 2’-hydroxy-3-phenylpropiophenone is a hydroxy- and phenyl-substituted propiophenone derivative with properties characteristic of hydroxyaryl ketones. It serves as an important intermediate in organic synthesis, with chemical reactivity influenced by its functional groups and substitution pattern. References 1987. Local anesthetic activity of derivatives of 2-hydroxy-3-phenylpropiophenone. Pharmaceutical Chemistry Journal, 21(9). DOI: 10.1007/bf01145557 2001. Microcolumn High-Performance Liquid Chromatography of ortho-Substituted Hydrophobic Phenol Derivatives. Journal of Analytical Chemistry, 56(4). DOI: 10.1023/a:1016600228992 2000. A Method for the Synthesis of 2,3-Disubstituted 2,3-Dihydrobenzofurans. Monatshefte für Chemie / Chemical Monthly, 131(1). DOI: 10.1007/s007060050318 |
| Market Analysis Reports |