Online Database of Chemicals from Around the World
| Xinlian Rui Biomedical Technology (shandong) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xlrchem.com | |||
 | +86 15153173989 | |||
 | 3865799121@qq.com | |||
 | QQ Chat | |||
 | WeChat: 15153173989 | |||
| Chemical manufacturer since 2024 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Organic raw materials >> Amino compound >> Amide compound |
|---|---|
| Name | 2,2-Difluoroacetamide |
| Molecular Structure | 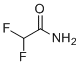 |
| Molecular Formula | C2H3F2NO |
| Molecular Weight | 95.05 |
| CAS Registry Number | 359-38-6 |
| EC Number | 818-090-2 |
| SMILES | C(C(=O)N)(F)F |
| Density | 1.3±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 46 - 50 °C (Expl.) |
| Boiling point | 231.7±25.0 °C 760 mmHg (Calc.)* |
| Flash point | 93.9±23.2 °C (Calc.)* |
| Index of refraction | 1.334 (Calc.)* |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
2,2-Difluoroacetamide is an organofluorine compound with the chemical formula CHF2CONH2. It consists of an acetamide backbone in which two hydrogen atoms on the α-carbon have been replaced with fluorine atoms. This substitution introduces strong electron-withdrawing effects, which significantly modify the compound’s acidity, hydrogen bonding, and chemical reactivity compared to unsubstituted acetamide. The compound was first synthesized during systematic investigations of fluorinated amides in the mid-20th century, when researchers explored how fluorine substitution influenced the physical and chemical properties of common functional groups. The difluoromethyl group (–CF2H) proved especially interesting due to its ability to mimic hydroxyl or thiol functionalities in biochemical contexts while maintaining high electronegativity and lipophilicity. 2,2-Difluoroacetamide is primarily applied as an intermediate in the preparation of pharmaceuticals, agrochemicals, and specialty fluorinated compounds. The presence of the difluoromethyl group makes it a useful building block for designing molecules with enhanced metabolic stability and altered biological activity. For example, in drug design, fluorinated amides can serve as bioisosteres that improve binding affinity to enzymes or receptors while resisting enzymatic degradation. In synthetic chemistry, 2,2-difluoroacetamide is employed in reactions that exploit both its amide functionality and the electron-withdrawing difluoromethyl group. It can undergo acylation, condensation, and substitution reactions, providing access to more complex fluorinated derivatives. Its reactivity profile makes it a useful starting material for introducing the difluoromethyl motif into heterocycles and other nitrogen-containing frameworks. The compound has also been investigated in biochemical studies because the difluoromethyl group can alter hydrogen-bonding interactions and polarity. Such properties are valuable in the design of enzyme inhibitors, where small changes in electronic distribution may lead to significant differences in activity and selectivity. Synthesis of 2,2-difluoroacetamide is typically achieved by converting difluoroacetic acid or its esters into the corresponding acid chloride, followed by treatment with ammonia to yield the amide. Alternatively, direct amidation methods using coupling reagents have also been reported to obtain the compound efficiently. Overall, 2,2-difluoroacetamide represents an important fluorinated building block in organic and medicinal chemistry. Its discovery and development reflect the broader role of fluorine chemistry in creating molecules with unique reactivity and properties that are highly valuable in both fundamental research and applied technologies. References 2024. Recent progress in carbene-catalyzed fluoroalkylation. Science China Chemistry, 67(7). DOI: 10.1007/s11426-024-1981-1 2023. The HBV capsid modulators derived from sulfamoylbenzamides and benzamides: an overview of the progress of patents. Medicinal Chemistry Research, 32(7). DOI: 10.1007/s00044-023-03095-x 2023. A gradient solid electrolyte interphase with high Li+ conductivity induced by bisfluoroacetamide additive for stable lithium metal batteries. Nano Research, 16(5). DOI: 10.1007/s12274-022-5363-6 |
| Market Analysis Reports |