Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Biotalk Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.biotalk.cn | |||
 | +86 (21) 6501-2399 6504-0613 | |||
 | +86 (21) 6501-8759 | |||
 | sales@biotalk.cn | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Weihai Newera Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.newerachem.com | |||
 | +86 (631) 592-0703 +86 13863171217 | |||
 | +86 (631) 592-0704 | |||
 | wenqingli@263.net wenqing-li@hotmail.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Win-Win Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.win-winchemical.com | |||
 | +86 (577) 6449-8589 +86 15325081899 | |||
 | +86 (577) 5699-4596 | |||
 | sales@win-winchemical.com winwinchemical@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Shanghai Hohance Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hohance.com | |||
 | +86 (21) 3111-5312 | |||
 | +86 (21) 3111-5317 | |||
 | info@hohance.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| SynQuest Labs, Inc. | USA | |||
|---|---|---|---|---|
 | www.synquestlabs.com | |||
 | +1 (386) 462-0788 | |||
 | +1 (386) 462-7097 | |||
 | Sales@synquestlabs.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Suli Pharmaceutical Technology Jiangyin Co., Ltd., | China | |||
|---|---|---|---|---|
 | www.sulipharma.com | |||
 | +86 (21) 5197-2823 ex 8033 | |||
 | qian.wang@suli.com | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Jinan Langhua Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lhchem.cn | |||
 | +86 (531) 8590-6691 | |||
 | +86 (531) 8590-6692 | |||
 | 325912681@qq.com | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Allfluoro pharmaceutical co., Ltd. | China | |||
|---|---|---|---|---|
 | www.allfluoro.com | |||
 | +86 (21) 2613-7118 | |||
 | sales@allfluoro.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Sifluorotm Co,. Ltd. | China | |||
|---|---|---|---|---|
 | www.fluoro.com | |||
 | +86 (0531) 8590-6692 | |||
 | +86 (0531) 8590-6692 | |||
 | inquiry@fluoro.com | |||
 | WeChat: 15288857388 | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Alfa Aesar. | USA | |||
|---|---|---|---|---|
 | www.alfa.com | |||
 | +1 (978) 521-6300 | |||
 | +1 (978) 521-6350 | |||
 | info@alfa.com | |||
| Chemical manufacturer | ||||
| P and M Invest Ltd. | Russia | |||
|---|---|---|---|---|
 | www.fluorine.ru | |||
 | +7 (495) 135-6494 | |||
 | +7 (495) 135-6509 | |||
 | sales@fluorine1.ru | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Chemical reagent >> Organic reagent >> Epoxide |
|---|---|
| Name | 1,1,1-Trifluoro-2,3-epoxypropane |
| Synonyms | 3,3,3-Trifluoro-1,2-epoxypropane |
| Molecular Structure | 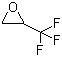 |
| Molecular Formula | C3H3F3O |
| Molecular Weight | 112.05 |
| CAS Registry Number | 359-41-1 |
| EC Number | 609-196-8 |
| SMILES | C1C(O1)C(F)(F)F |
| Density | 1.4±0.1 g/cm3, Calc.*, 1.3 g/mL (Expl.) |
|---|---|
| Index of Refraction | 1.326, Calc.*, 1.297 (Expl.) |
| Boiling Point | 33.5±35.0 °C (760 mmHg), Calc.*, 38-40 °C (Expl.) |
| Flash Point | -21.3±21.8 °C, Calc.*, -40 °C (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS02;GHS07 Warning Details GHS02;GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H225-H302-H312-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P303+P361+P353-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P370+P378-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||
|
1,1,1-Trifluoro-2,3-epoxypropane (C3H3F3O) is a fluorinated epoxy compound, containing both an epoxide group and three fluorine atoms attached to the first carbon of the propane chain. This compound belongs to a class of fluorinated organic compounds that have been studied for their unique properties derived from the presence of fluorine atoms. Fluorinated molecules, in general, exhibit enhanced chemical stability, resistance to solvents, and high thermal stability, which makes them valuable in various chemical applications. The compound was first synthesized as part of ongoing research into fluorinated epoxy compounds and their potential uses in organic synthesis and material science. The incorporation of fluorine into epoxy compounds, which are typically highly reactive and used for curing and polymerization processes, has been of particular interest for the development of novel materials with specialized properties. 1,1,1-Trifluoro-2,3-epoxypropane is primarily utilized as a reactive intermediate in chemical synthesis. Its epoxy group is highly reactive and can participate in nucleophilic substitution reactions, making it a valuable building block for the synthesis of various fluorinated compounds. The presence of the trifluoromethyl group on the carbon chain enhances the reactivity of the epoxide group, providing opportunities for the formation of more complex fluorinated molecules. One of the main applications of 1,1,1-trifluoro-2,3-epoxypropane is in the preparation of fluorinated polymers. Fluorinated polymers are known for their high resistance to heat, chemical solvents, and electrical conductivity, which makes them useful in a variety of high-performance materials. By incorporating 1,1,1-trifluoro-2,3-epoxypropane as a monomer or a precursor in the polymerization process, researchers can tailor the properties of the resulting polymer to meet specific needs, particularly in industries such as electronics, aerospace, and automotive manufacturing. The compound has also found use in the preparation of fluorinated surfactants and coatings. Fluorinated surfactants, which are used to reduce surface tension and improve wetting properties, are valuable in industries such as coatings, detergents, and oil recovery. The trifluoromethyl group in 1,1,1-trifluoro-2,3-epoxypropane can be used to modify the surface characteristics of materials, making them more resistant to water and oil, which is important in various industrial processes. In addition, 1,1,1-trifluoro-2,3-epoxypropane has been explored for its potential use in pharmaceuticals and agrochemicals, as fluorinated compounds are known to exhibit unique biological activities and stability. The compound's ability to undergo chemical reactions that lead to the formation of more complex fluorinated structures has made it a valuable intermediate in the development of fluorinated drugs and crop protection agents. Due to the presence of fluorine, which is known for its environmental persistence, the use and disposal of 1,1,1-trifluoro-2,3-epoxypropane must be carefully managed. Fluorinated compounds are resistant to biodegradation and can accumulate in the environment if not handled properly. Consequently, regulations regarding the environmental impact of fluorinated chemicals require that appropriate measures be taken to prevent contamination and ensure safe disposal. In conclusion, 1,1,1-trifluoro-2,3-epoxypropane is an important fluorinated chemical compound with a wide range of applications in organic synthesis, polymer science, and materials engineering. Its unique combination of an epoxy group and fluorine atoms makes it a versatile building block for the development of high-performance materials, surfactants, and pharmaceuticals. However, like other fluorinated compounds, its environmental and safety considerations demand careful management to minimize its impact on the ecosystem. References 2021. Site-specific interrogation of an ionic chiral fragment during photolysis using an X-ray free-electron laser. Communications Chemistry, 4(1). DOI: 10.1038/s42004-021-00555-6 2011. Preparation of Optically Pure α-Trifluoromethyl-α-amino Acids from N-Tosyl-2-trifluoromethyl-2-alkyloxycarbonyl Aziridine. The Journal of Organic Chemistry, 76(22). DOI: 10.1021/jo201554g 1969. Nature of the catalytic centers in the polymerization of trifluoropropylene oxide in the presence of ferric chloride. Bulletin of the Academy of Sciences of the USSR, Division of chemical science, 18(2). DOI: 10.1007/bf00905538 |
| Market Analysis Reports |