Online Database of Chemicals from Around the World
| Shanghai Techwell Biopharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.techwell-cn.com | |||
 | +86 (21) 5483-1973 | |||
 | +86 (21) 2301-0563 | |||
 | sales@techwell-cn.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Manus Aktteva | India | |||
|---|---|---|---|---|
 | www.manusaktteva.in | |||
 | +91 (79) 6512-3395 | |||
 | +91 (79) 2646-3395 | |||
 | products@manusakttevabiopharma.in | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2008 | ||||
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Zhejiang Ausun Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ausunpharm.com | |||
 | +86 (576) 8558-9335 +86 13957690608 | |||
 | +86 (576) 8558-9168 | |||
 | sales@ausunpharm.com | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Chirogate International Inc. | Taiwan | |||
|---|---|---|---|---|
 | www.chirogate.com | |||
 | +886 (3) 496-3808 | |||
 | +886 (3) 496-3800 | |||
 | annyeh@chirogate.com kirogate@ms63.hinet.net | |||
| Chemical manufacturer since 2000 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Taizhou KEDE Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kedechemical.com | |||
 | +86 (576) 8461-3060 +86 13093829633 | |||
 | +86 (576) 8461-3060 | |||
 | sales@kedechemical.com | |||
 | QQ Chat | |||
 | WeChat: 13093829633 | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Shanghai Genriver Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.genriverpharm.com | |||
 | +86 13761582449 +86 13482015261 | |||
 | +86 (21) 3778-2903 | |||
 | info@genriverpharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Tecoland Corporation | USA | |||
|---|---|---|---|---|
 | www.tecoland.com | |||
 | +1 (732) 603-9577 | |||
 | +1 (732) 906-1522 | |||
 | info@tecoland.com | |||
| Chemical distributor since 2001 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Taizhou Runsun Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.runsunchemical.com | |||
 | +86 15867635987 | |||
 | sales@runsunchemical.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Finetech Industry Limited | China | |||
|---|---|---|---|---|
 | www.finetechchem.com | |||
 | +86 (27) 8746-5837 +86 18971612321 | |||
 | +86 (27) 8777-2287 | |||
 | sales@finetechnology-ind.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Cayman Chemical Company | USA | |||
|---|---|---|---|---|
 | www.caymanchem.com | |||
 | +1 (734) 971-3335 | |||
 | +1 (734) 971-3640 | |||
 | sales@caymanchem.com | |||
| Chemical manufacturer | ||||
| Tocris Bioscience Inc. | USA | |||
|---|---|---|---|---|
 | www.tocris.com | |||
 | +1 (636) 207-7651 | |||
 | +1 (636) 207-7683 | |||
 | customerservice.na@bio-techne.com | |||
| Chemical manufacturer since 1982 | ||||
| Classification | API >> Hormone and endocrine-regulating drugs >> Prostaglandins |
|---|---|
| Name | Prostaglandin E2 |
| Synonyms | Dinoprostone; (5Z,11a,13E,15S)-11,15-Dihydroxy-9-oxoprosta-5,13-dien-1-oic acid; 7-[3-Hydroxy-2-(3-hydroxy-1-octenyl)-5-oxocyclopentyl]-5-heptenoic acid; PGE2 |
| Molecular Structure | 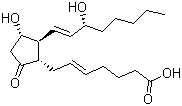 |
| Molecular Formula | C20H32O5 |
| Molecular Weight | 352.47 |
| CAS Registry Number | 363-24-6 |
| EC Number | 206-656-6 |
| SMILES | CCCCC[C@@H](/C=C/[C@H]1[C@@H](CC(=O)[C@@H]1C/C=CCCCC(=O)O)O)O |
| Density | 1.1±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 66 °C (Expl.) |
| Boiling point | 530.1±50.0 °C 760 mmHg (Calc.)* |
| Flash point | 288.5±26.6 °C (Calc.)* |
| Solubility | water insoluble, DMSO (100mM), ethanol (45mM) (Expl.) |
| Index of refraction | 1.561 (Calc.)* |
| Alpha | -85.5 ° (c=1, C2H5OH) (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS08 Danger Details GHS07;GHS08 Danger Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H360 Details | ||||||||||||||||||||||||
| Safety Statements | P203-P264-P270-P280-P301+P317-P318-P330-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
Prostaglandin E2 (PGE2) is a naturally occurring eicosanoid derived from arachidonic acid through the cyclooxygenase (COX) pathway. It was first identified in human seminal plasma in the 1930s and subsequently characterized as a prostaglandin with potent biological activity in multiple tissues. PGE2 is synthesized by the sequential action of COX enzymes, which convert arachidonic acid to the unstable intermediate prostaglandin H2 (PGH2), followed by PGE synthases that catalyze the formation of PGE2. PGE2 functions as a local autocrine and paracrine signaling molecule due to its rapid metabolism and short half-life. PGE2 exerts its effects through binding to four distinct G protein-coupled receptors, designated EP1, EP2, EP3, and EP4, each of which mediates different intracellular signaling pathways. EP1 typically activates phospholipase C and increases intracellular calcium, EP2 and EP4 activate adenylate cyclase to raise cyclic AMP levels, and EP3 couples to multiple G proteins to produce inhibitory or stimulatory effects depending on the tissue context. The tissue-specific distribution of these receptors explains the diverse physiological and pathophysiological roles of PGE2. Physiologically, PGE2 regulates vascular tone, gastrointestinal integrity, renal function, bone remodeling, and reproductive processes. In the gastrointestinal tract, it maintains mucosal protection by stimulating mucus and bicarbonate secretion and promoting mucosal blood flow. In the kidneys, it modulates glomerular filtration and sodium excretion. PGE2 also plays a critical role in inflammation and fever, mediating vasodilation, increasing vascular permeability, and acting on the hypothalamus to induce pyrexia. Furthermore, PGE2 is involved in labor induction by promoting cervical ripening and uterine contractions, which has led to its clinical application as a labor-inducing agent. The clinical applications of PGE2 include the induction of labor, termination of pregnancy, treatment of gastric ulcers, and the management of patent ductus arteriosus in neonates. Synthetic analogs and stable derivatives have been developed to enhance stability, receptor selectivity, and therapeutic efficacy. Misoprostol, a methyl ester analog of PGE1, and dinoprostone, the pharmacologically active form of PGE2, are examples used in obstetrics and gynecology to induce labor or as abortifacients. These derivatives exploit the receptor-mediated actions of PGE2 while improving pharmacokinetic properties such as oral bioavailability and metabolic stability. Research on PGE2 has also highlighted its involvement in pathological conditions, including cancer, chronic inflammation, and cardiovascular diseases. Overexpression of COX-2 and elevated PGE2 levels are associated with tumorigenesis, angiogenesis, and immune modulation in cancer. PGE2 contributes to inflammatory disorders such as rheumatoid arthritis by promoting synovial inflammation and pain. These findings have spurred the development of COX inhibitors and EP receptor modulators as therapeutic strategies to regulate PGE2 signaling in disease. References Funk CD (2001) Prostaglandins and leukotrienes: advances in eicosanoid biology. Science 294 1871–1875 DOI: 10.1126/science.294.5548.1871 Ricciotti E, FitzGerald GA (2011) Prostaglandins and inflammation. Arteriosclerosis, Thrombosis, and Vascular Biology 31 986–1000 DOI: 10.1161/ATVBAHA.110.207449 Regan JW (2003) EP2 and EP4 prostanoid receptor signaling. Life Sciences 74 143–153 DOI: 10.1016/j.lfs.2003.09.031 |
| Market Analysis Reports |