Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Ich Biofarm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ichemie.com | |||
 | +86 (571) 2818-6870 | |||
 | +86 (571) 8993-9479 | |||
 | specialchem1@ichemie.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Venkateshwara Life Sciences | India | |||
|---|---|---|---|---|
 | www.venkateshwaralifesciences.com | |||
 | +91 (9963) 894-542 | |||
 | balakrishna@venkateshwaralifesciences.com | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Agrige Bioscience Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.agrige.com | |||
 | +86 17321027985 | |||
 | sales@agrige.com | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Classification | Chemical reagent >> Organic reagent >> Fatty aldehyde (containing acetal, hemiacetal) |
|---|---|
| Name | 2-Chloro-1,3-propanedial |
| Synonyms | Chloromalonaldehyde; Chloromalondialdehyde; Chloromalonic dialdehyde |
| Molecular Structure | 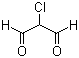 |
| Molecular Formula | C3H3ClO2 |
| Molecular Weight | 106.51 |
| CAS Registry Number | 36437-19-1 |
| EC Number | 689-033-5 |
| SMILES | C(=O)C(C=O)Cl |
| Density | 1.4±0.1 g/cm3 Calc.*, 1.261 g/mL (Expl.) |
|---|---|
| Melting point | 145-146 °°C (dec.) (Expl.) |
| Boiling point | 204.6±40.0 °C 760 mmHg (Calc.)* |
| Flash point | 77.5±27.3 °C (Calc.)* |
| Index of refraction | 1.489 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
2-Chloro-1,3-propanedial is a bifunctional organic compound characterized by the presence of two aldehyde groups at the 1 and 3 positions of a three-carbon chain and a chlorine atom substituent at the 2 position. Its molecular formula is C3H3ClO2, reflecting the combination of carbon, hydrogen, chlorine, and oxygen atoms. The molecule is structurally an aldehyde derivative of chlorinated propane with reactive functionalities that enable a wide range of chemical transformations. This compound’s two aldehyde groups confer significant electrophilic character, allowing it to participate readily in nucleophilic addition and condensation reactions. The positioning of these aldehyde groups at terminal carbons and the chlorine atom at the central carbon creates a molecule with both reactive centers and potential sites for substitution or elimination reactions. The presence of the chlorine atom also introduces the possibility of nucleophilic substitution at the 2-position, providing synthetic versatility. Synthetically, 2-chloro-1,3-propanedial can be prepared by the chlorination of glyceraldehyde derivatives or through oxidative halogenation of chloropropanol precursors. The compound may be isolated under controlled conditions due to its high reactivity and potential instability caused by the proximity of the reactive aldehyde groups and halogen substituent. In organic synthesis, 2-chloro-1,3-propanedial is valuable as a building block for the preparation of heterocycles, pharmaceuticals, and agrochemicals. Its dialdehyde functionality allows it to form cyclic structures through intramolecular or intermolecular condensation reactions, while the chlorine substituent can be replaced or eliminated to introduce further diversity. For example, it can be used to synthesize pyrrole, furan, or other nitrogen- or oxygen-containing heterocycles via condensation with amines or hydroxyl compounds. The compound's reactivity also makes it useful in the modification of biomolecules and polymers. Its aldehyde groups can form Schiff bases with amino groups, enabling covalent attachment to proteins, peptides, or other amine-containing substrates. This property is exploited in biochemical labeling and cross-linking applications. Physicochemically, 2-chloro-1,3-propanedial is typically a reactive liquid or low-melting solid. It is soluble in polar organic solvents but may undergo polymerization or degradation if not handled under appropriate conditions. Analytical characterization includes nuclear magnetic resonance spectroscopy, which reveals the aldehyde proton signals, and infrared spectroscopy, where strong absorption bands near 1720 cm-1 correspond to the carbonyl stretching vibrations. Safety considerations are important when handling 2-chloro-1,3-propanedial due to its electrophilic and potentially irritant nature. Appropriate protective measures should be employed to prevent inhalation, skin contact, or exposure to reactive intermediates formed during its reactions. In summary, 2-chloro-1,3-propanedial is a highly reactive chlorinated dialdehyde with applications in synthetic organic chemistry, biochemical modification, and the preparation of heterocyclic compounds. Its bifunctional aldehyde groups and halogen substituent offer a versatile platform for chemical transformations and molecular construction in various fields. References 2023. Antituberculotic Activity of 5-Aryl(Hetaryl)-Methylidene- 2,4,6-Pyrimidine-2,4,6(1H,3H,5H)-Triones and 5-(2-Chloropropylidene)-2,4,6-Pyrimidine-2,4,6(1H,3H,5H)-Triones. Pharmaceutical Chemistry Journal. DOI: 10.1007/s11094-023-02885-2 2020. Synthesis and Antimycobacterial Activity of 5-(Hetarylmethylidene)-2,4,6-Pyrimidine- 2,4,6(1H,3H,5H )-Triones And 5-(2-Chloropropylidene)- 2,4,6-Pyrimidine-2,4,6(1H,3H,5H )-Triones. Pharmaceutical Chemistry Journal. DOI: 10.1007/s11094-020-02169-z 2006. Time-resolved photoelectron spectroscopy of proton transfer in the ground state of chloromalonaldehyde: Wave-packet dynamics on effective potential surfaces of reduced dimensionality. The Journal of Chemical Physics, 124(15). DOI: 10.1063/1.2191852 |
| Market Analysis Reports |