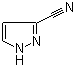
3‑Cyanopyrazole (also called 1H‑pyrazole‑3‑carbonitrile) is a small heterocyclic nitrile with the molecular formula C4H3N3 and CAS number 36650‑74‑5. The pyrazole ring includes two adjacent nitrogen atoms, and the cyano (–C≡N) group is attached at the 3‑position, making this compound a useful intermediate in heterocyclic and medicinal chemistry.
This molecule has been used as a building block in the synthesis of more complex pyrazole derivatives. Because of its cyano group, it is amenable to a variety of chemical transformations: it can be converted into amidines, amides, or other nitrogen‑containing functional groups, or participate in cycloaddition reactions. The pyrazole core itself is a privileged scaffold in drug discovery, and addition of a nitrile moiety increases its utility, enabling the design of molecules that can serve as enzyme inhibitors, ligands, or biologically active heterocycles.
In synthetic chemistry practice, 3‑cyano‑pyrazole can be used in palladium‑catalyzed cross‑couplings, in addition to nucleophilic attacks on the nitrile to build up molecular complexity. Its relative stability and manageable reactivity make it a good substrate for multistep organic synthesis. Because the molecule is fairly simple and commercially available in high purity, it is commonly employed in medicinal chemistry as a starting point for hit‑to‑lead optimization.
As a solid compound, 3‑cyanopyrazole requires standard laboratory precautions: it should be stored in a dry environment to avoid moisture uptake, and reactions involving the nitrile group are typically performed in aprotic solvents under inert atmosphere to prevent side-reactions. There is limited data on pharmacological properties for 3‑cyano‑pyrazole itself — most applications stem from its use as a synthetic building block rather than as a final bioactive agent.
Because of its versatility, 3‑cyanopyrazole remains a valuable intermediate in the synthesis of nitrogen-rich heterocycles, offering a bridge to more elaborated pyrazole-based frameworks in materials science, medicinal chemistry, and organic synthesis.
References
Synthesis and Antimicrobial Activity of Substituted Amino-1H-Pyrazoles (2025) Pharmaceutical Chemistry Journal DOI: 10.1007/s11094-025-03358-4
Reactions of spirocyclopropane-containing 1- and 2-pyrazolines with electrophilic reagents (2000) Russian Chemical Bulletin 49 3 601–607 DOI: 10.1007/bf02494777
Synthesis and Pharmacological Activities of Pyrazole and Oxadiazole Derivatives: a Review (2023) Russian Journal of Organic Chemistry 59 5 732–752 DOI: 10.1134/s1070428023050160
|





















 GHS07 Warning Details
GHS07 Warning Details