N,N-Diethylhydroxylamine is an organic compound with the formula C4H11NO. It is widely used as a reducing agent and an antioxidant in various chemical applications. Discovered in the early 20th century, it has since gained importance in industrial processes, especially in polymer stabilization and lubrication. As a reducing agent, N,N-diethylhydroxylamine is involved in numerous organic syntheses, where its ability to donate electrons makes it valuable in reducing nitro compounds to amines. Over the years, its use has expanded to include roles in polymer chemistry, metal ion detection, and food preservation, among others.
The compound was first recognized for its usefulness in preventing oxidation in synthetic materials. One of its key applications is in stabilizing polyvinyl chloride (PVC) during its production, where it functions as a radical scavenger, protecting the polymer from heat and UV degradation. This made N,N-diethylhydroxylamine essential for the manufacture of durable plastics and other materials sensitive to environmental factors. In addition, the substance is used in the creation of synthetic lubricants, where it prevents the oxidation of oils and greases, ensuring long-term performance under high-stress conditions.
N,N-Diethylhydroxylamine is also employed in chemical synthesis as a reducing agent. It is frequently used to reduce nitro compounds to amines, a crucial step in the production of pharmaceuticals and agrochemicals. Its reducing properties extend to other applications, such as in the preparation of hydrazine derivatives, which are used in rocket fuels and other high-energy applications. This versatility makes the compound an essential tool in the chemical industry, providing efficient solutions in both laboratory and large-scale manufacturing settings.
In addition to its industrial uses, N,N-diethylhydroxylamine has been employed in analytical chemistry. It is utilized in specific assays designed to detect metal ions, such as copper and iron, which are important in various biochemical and environmental studies. Furthermore, the compound has been explored for its potential in the preservation of food products. By acting as an antioxidant, it helps to extend the shelf life of food and beverages by preventing oxidation, which can lead to spoilage.
However, the widespread use of N,N-diethylhydroxylamine has raised concerns about its potential environmental and health impacts. Studies have investigated the compound’s biodegradability and its effects on aquatic life, as its use in industrial processes could lead to environmental contamination. Efforts are being made to find safer alternatives and to improve the application methods of N,N-diethylhydroxylamine to mitigate any adverse effects.
Despite these concerns, N,N-diethylhydroxylamine continues to be a valuable compound in a variety of industries, from polymer production to environmental chemistry. Ongoing research into its safety and efficacy is crucial to ensure that it remains a reliable and sustainable option for chemical processes worldwide.
References
2024. Effect of monomethylhydrazine on γ-rays radiolysis and radiolytic by-products of N,N-diethylhydroxylamine in nitric acid solution. Journal of Radioanalytical and Nuclear Chemistry, 333(8).
DOI: 10.1007/s10967-024-09661-w
2023. Triethylamine-Promoted Oxidative Cyclodimerization of 2H-Azirine-2-carboxylates to Pyrimidine-4,6-dicarboxylates: Experimental and DFT Study. Molecules, 28(11).
DOI: 10.3390/molecules28114315
2022. Spectroscopic and quantum mechanical study of a scavenger molecule: N,N-diethylhydroxylamine. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 281.
DOI: 10.1016/j.saa.2022.121555
|






























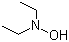


 GHS02;GHS07;GHS09 Warning Details
GHS02;GHS07;GHS09 Warning Details