Online Database of Chemicals from Around the World
| Epochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.epochem.com | |||
 | +86 (21) 6760-1595 6760-1597 | |||
 | +86 (21) 6760-1605 | |||
 | seth_wang@epochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Fluoropharm Co.,Ltd. | China | |||
|---|---|---|---|---|
 | www.fluoropharm.com | |||
 | +86 (571) 8558-6753 +86 13336034509 | |||
 | +86 (571) 8558-6753 | |||
 | sales@fluoropharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2013 | ||||
| Shandong Huimeng Biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.huimengchem.com | |||
 | +86 (530) 884-2888 +86 18523002917 | |||
 | +86 (530) 884-2666 | |||
 | cindy@szgoldenunion.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Hangzhou Molcore Biopharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.molcore.com | |||
 | +86 (571) 8102-5280 | |||
 | sales@molcore.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Ricci Chimica | Italy | |||
|---|---|---|---|---|
 | www.riccichimica.com | |||
 | +39 (75) 692-9241 | |||
 | +39 (75) 592-6595 | |||
 | info@riccichimica.com | |||
| Chemical manufacturer | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridine compound >> Methylpyridine |
|---|---|
| Name | 3-Trifluoromethylpyridine |
| Synonyms | 3-(Trifluoromethyl)pyridine |
| Molecular Structure | 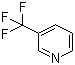 |
| Molecular Formula | C6H4F3N |
| Molecular Weight | 147.10 |
| CAS Registry Number | 3796-23-4 |
| EC Number | 223-268-2 |
| SMILES | C1=CC(=CN=C1)C(F)(F)F |
| Density | 1.276 |
|---|---|
| Boiling point | 113-115 °C |
| Refractive index | 1.4173 |
| Flash point | 23 °C |
| Hazard Symbols |    GHS02;GHS06;GHS07 Danger Details GHS02;GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H225-H226-H300-H315-H319-H335 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P261-P264-P264+P265-P270-P271-P280-P301+P316-P302+P352-P303+P361+P353-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P370+P378-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||
|
3-Trifluoromethylpyridine, also known as 3-(trifluoromethyl)pyridine, is a valuable compound characterized by a trifluoromethyl group at the third position of the pyridine ring. Its chemical formula is C6H4F3N. Due to its unique chemical properties, this compound has attracted great interest in various fields including pharmaceuticals, agrochemicals, and organic synthesis. The discovery of 3-trifluoromethylpyridine stems from the extensive exploration of fluorine-containing organic compounds in the mid-20th century. Due to its high electronegativity and small size, fluorine can significantly change the physical, chemical, and biological properties of organic molecules. The trifluoromethyl group with three fluorine atoms is particularly effective in enhancing the stability, lipophilicity, and metabolic resistance of the compound. Chemists synthesized 3-trifluoromethylpyridine as part of an effort to exploit these advantages for various applications. In the pharmaceutical industry, 3-trifluoromethylpyridine is a key building block. The presence of trifluoromethyl groups in drug molecules can improve their binding affinity to biological targets, increase metabolic stability, and improve bioavailability. These properties are essential for the design of effective drugs. As a result, 3-trifluoromethylpyridine is used to synthesize a variety of pharmaceutical agents, including those targeting neurological disorders, cardiovascular disease, and cancer. Its role as a synthetic intermediate enables medicinal chemists to develop drugs with improved efficacy and safety. The agrochemical industry also benefits from the unique properties of 3-trifluoromethylpyridine. It is used in the synthesis of herbicides, fungicides, and insecticides. The trifluoromethyl group helps improve the potency and environmental persistence of these agrochemicals, ensuring effective control of pests and weeds. Furthermore, the addition of the fluorinated group helps reduce the likelihood of pests developing resistance, making 3-trifluoromethylpyridine an important component in sustainable agricultural practices. In organic synthesis, 3-trifluoromethylpyridine is highly regarded for its versatility and reactivity. The electron-withdrawing trifluoromethyl group affects the electronic properties of the pyridine ring, making it a valuable intermediate for a wide range of chemical reactions. It is particularly useful in cross-coupling reactions, nucleophilic substitutions, and other transformations necessary to build complex organic molecules. This versatility supports the synthesis of fine chemicals, specialty materials, and advanced polymers, expanding the scope of chemical research and industrial applications. Standard laboratory precautions are required for handling and storage of 3-trifluoromethylpyridine. The compound should be stored in a cool, dry place away from incompatible materials. During handling, appropriate safety precautions must be taken, including the use of gloves, goggles, and adequate ventilation to prevent contact. In addition, proper disposal methods should be followed to minimize environmental impact and ensure safety. References 2014. Synthesis of Fluorinated Pyridines. Fluorine in Heterocyclic Chemistry Volume 2. DOI: 10.1007/978-3-319-04435-4_1 [No volume/issue available] 2013. Reactions of Pyridines, Benzopyridines, and Azapyridines with Organomagnesiums and Organolithiums. Metalation of Azines and Diazines. DOI: 10.1007/7081_2012_96 [No volume/issue available] 2012. Recent Developments on the Trifluoromethylation of (Hetero)Arenes. Chemistry - An Asian Journal, 7(7). DOI: 10.1002/asia.201200211 |
| Market Analysis Reports |