Online Database of Chemicals from Around the World
| Beijing Eagle Sky Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.eagleskypharmatech.com | |||
 | +86 (10) 5979-9429 8875-5821 | |||
 | +86 (10) 5804-3698 | |||
 | sophia_818@126.com contact@eagleskypharmatech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Premium supplier since 2010 | ||||
| Classification | Organic raw materials >> Amino compound >> Acyclic monoamines, polyamines and their derivatives and salts |
|---|---|
| Name | (R)-(+)-1-(1-Naphthyl)ethylamine |
| Synonyms | R-(+)-alpha-(1-Naphthyl)ethylamine |
| Molecular Structure | 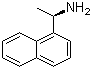 |
| Molecular Formula | C12H13N |
| Molecular Weight | 171.24 |
| CAS Registry Number | 3886-70-2 |
| EC Number | 223-425-5 |
| SMILES | C[C@H](C1=CC=CC2=CC=CC=C21)N |
| Density | 1.067 g/mL (Expl.) |
|---|---|
| Boiling point | 153 °C (11 mmHg) (Expl.) |
| Refractive index | 1.623 (Expl.) |
| alpha | 55 ° (c=2, CH3OH) (Expl.) |
| Flash point | 113 °C (Expl.) |
| Hazard Symbols |     GHS05;GHS06;GHS07;GHS09 Danger Details GHS05;GHS06;GHS07;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H314-H315-H318-H319-H335-H411 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P261-P264-P264+P265-P270-P271-P273-P280-P301+P316-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P351+P338-P305+P354+P338-P316-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P363-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||
|
(R)-(+)-1-(1-Naphthyl)ethylamine is a chiral amine characterized by the presence of a naphthyl group attached to a secondary amine, which makes it an important building block in asymmetric synthesis. This compound is a key example of the utilization of stereochemical principles in modern organic and pharmaceutical chemistry. Its discovery stems from efforts to identify chiral auxiliaries and ligands that enable enantioselective transformations, particularly in the synthesis of biologically active molecules. The structure of (R)-(+)-1-(1-Naphthyl)ethylamine confers unique properties that make it highly versatile. The presence of the rigid naphthyl moiety ensures a defined spatial orientation, while the chiral center at the ethylamine linkage introduces asymmetry essential for enantioselective interactions. These properties make the compound a preferred choice in catalytic and stoichiometric applications in organic synthesis. A major application of (R)-(+)-1-(1-Naphthyl)ethylamine is its role as a chiral auxiliary in asymmetric synthesis. It is often used to introduce chirality into target molecules through the formation of diastereomeric intermediates. For example, it has been employed in the stereoselective synthesis of amino acids, alkaloids, and other complex natural products. Its ability to influence reaction outcomes is attributed to the steric and electronic effects imparted by the naphthyl and ethylamine groups. The compound is also widely used in the pharmaceutical industry as a precursor for the synthesis of active pharmaceutical ingredients (APIs). It serves as a chiral building block in the production of enantiomerically pure drugs. The pharmaceutical relevance of (R)-(+)-1-(1-Naphthyl)ethylamine is further underscored by its use in the preparation of ligands and catalysts for enantioselective transformations, enabling efficient and sustainable drug manufacturing processes. Another significant application is in materials science, where (R)-(+)-1-(1-Naphthyl)ethylamine is used to synthesize optically active polymers and supramolecular assemblies. Its chiral properties contribute to the development of materials with specific optical and mechanical characteristics, enhancing their application in areas such as liquid crystals, sensors, and advanced coatings. In addition to its synthetic applications, the compound is a valuable tool in analytical chemistry. It is employed as a resolving agent for racemic mixtures, enabling the separation of enantiomers based on differential interactions with the chiral amine. This utility is critical for the analysis and characterization of chiral substances in both research and industrial contexts. Overall, the discovery and development of (R)-(+)-1-(1-Naphthyl)ethylamine highlight the interplay between stereochemistry and functionality in advancing chemical sciences. Its versatility in synthesis, pharmaceuticals, and materials science underscores its enduring importance in a variety of fields, making it a cornerstone of chiral chemistry. References 2022. Transaminase-mediated chiral selective synthesis of (1R)-(3-methylphenyl)ethan-1-amine from 1-(3-methylphenyl)ethan-1-one. Bioprocess and Biosystems Engineering. DOI: 10.1007/s00449-022-02824-7 2023. Direct syntheses of stereoisomers of 3-fluoro GABA and beta-fluoroamine analogues of the calcium receptor (CaR) agonists. Medicinal Chemistry Research. DOI: 10.1007/s00044-023-03103-0 |
| Market Analysis Reports |